Diabetes
Jonas de Jong and Alice Li
Preamble
Diabetes (diabetes mellitus) is one of the metabolic diseases with higher blood sugar level, either due to the pancreatic beta cells do not produce enough insulin, or the cells do not respond to the insulin that is produced.[1] Its clinical symptoms include three polies: polyuria (frequent urination), polydipsia (increased thirst) and polyphagia (increased hunger).
Diabetes is categorized as three types in the clinic: Type 1 diabetes (T1D, diabetes mellitus 1, DM1) results from pancreatic beta cell destruction and thus fails to produce insulin, and is insulin dependent which relies on insulin injection, it is also named insulin-dependent diabetes mellitus (IDDM). Since T1D happens mostly in juveniles it is also called juvenile-onset diabetes. Type 2 diabetes (T2D, diabetes mellitus 2, DM2) results from the reason that the beta cells do not respond to insulin (insulin resistance), and it may co-exist with the situation of partially or fully insulin-dependent, which is also named non-insulin-dependent diabetes mellitus (NIDDM) or adult-onset diabetes. In some type 2 diabetes, concomitant insulin therapy may be necessary (IDDM2). The third type is gestational diabetes occurs while pregnancy.
| Comparison of type 1 and 2 diabetes[2] | ||
|---|---|---|
| Feature | Type 1 diabetes | Type 2 diabetes |
| Onset | Sudden | Gradual |
| Age at onset | Mostly in children | Mostly in adults |
| Body habitus | Thin or normal[3] | Often obese |
| Ketoacidosis | Common | Rare |
| Autoantibodies | Usually present | Absent |
| Endogenous insulin | Low or absent | Normal, decreased or increased |
| Concordance in identical twins | 50% | 90% |
| Prevalence | ~10% | ~90% |
There is no cure for diabetes currently except that most of the gestational diabetes disappears when the pregnancy is ended, but it is treatable. The current therapies are insulin, other non-curing medications, and pancreatic replacement therapies like pancreatic islet transplantations which have been applied to severe T1D cases successfully since 1980s’.[4]Since both T1D and T2D are chronic diseases and the progresses of modern medicine, acute complications like hypoglycemia and ketoacidosis are under well controlled, and thus chronic and long-term complications like chronic renal failure, and diabetic retinopathy, especially cardiovascular diseases (CVD), are drawing attentions.
| Basic treatment steps of diabetes: | |
|---|---|
| Step 1 | Educate the patient
Treat concomittant risk factors (e.g. hypertension, obesity, hyperlipidemia) start met metformine (500 mg 1 dd, max. 1000 mg 3 dd) start cholesterol lowering drug (e.g. simvastatine 40mg once daily) start ACE inhibitor if no hypertension but with microalbuminuria (e.g. enalapril 10-20mg once daily) |
| Step 2 | BMI <27: add sulfonylureumderivate (eg. tolbutamide 500 mg once daily, max. 1000 mg twice daily)
BMI =27: add sulfonylureumderivaat if no CVD or heart failure If CVD but no increased risk of heart failure add (pioglitazon 15 mg once daily, max. 45 mg once daily) |
| Step 3 | Add long acting insulin to oral treatment (stop pioglitazon if present) |
| Step 4a | NPH-insuline or mix-insuline twice daily |
| Step 4b | Insulin four times daily |
Case Report
Rick is a 56-year-old plumber of a local firm, and his friends called him stocky since he is 7’8” and 260 lbs. He is married and was diagnosed with type 2 diabetes 14 months ago. He used to smoke 10 cigarettes a day and is currently not smoking, but he continued to eat fatty fast food seven to ten times per week due to his flow of work.
He had followed lifestyle advice and lost some weight and was doing more exercises, but had now been diagnosed with hypertension (163/95 mmHg). His physician decided to offer Rick some treatments for his hypertension, and Rick attended for a further check-up 18 months after the diabetic diagnosis. It was found that Rick now has chest pain, and he had to start taking aspirin and a beta blocker.
Introduction
As per the World Health Organization (WHO), an estimated 347 million people world-wide have diabetes in 2012.[5] The number of deaths attributed to diabetes was previously estimated at just over 800,000. However, it has long been known that the number of deaths related to diabetes is considerably underestimated. With regarding the causes of death of diabetes, one important factor contributing the increased morbidity and mortality in diabetic individuals is the development of cardiovascular disease, one of the chronic complications of diabetes. Several studies have demonstrated that diabetic patients have a risk of death that is two to three times higher than that among people without diabetes.[2]
| The following is a comprehensive list of other causes of diabetes:[6] | |
|---|---|
|
|
Multiple epidemiologic studies have established diabetes as a major risk factor for the development of all manifestations of cardiovascular disease, including myocardial infarction, stroke, peripheral vascular disease, and heart failure,[3][8][9][10] and recent data suggest that the proportion of cardiovascular disease attributable to diabetes is increasing.[8] It is estimated that cardiovascular disease accounts for 65% of all deaths in persons with diabetes.[11] In a recent meta-analysis of nearly 700,000 people from 102 prospective studies, diabetes conferred an approximate two fold risk for coronary heart disease and stroke, independently from other conventional risk factors.[10] Thus, in order to reduce the health burden of diabetes, it is considered necessary to aggressively prevent and treat cardiovascular disease in these patients.
Diabetes and pre-diabetic glucose abnormalities
Type 1 diabetes is caused by the gradually decreasing of endogenous insulin production by pancreatic beta cells and thus loss of the control of blood glucose, whereas type 2 diabetes is caused by the rising blood glucose resulted from a combination of genetic predisposition, unhealthy diet, physical inactivity, and body weight-gain.[12] The higher than normal blood glucose level in both type 1 and type 2 diabetes results in pathophysiological processes, which are associated with the development of microvascular disease and atherosclerosis. Patients with diabetes are thus at a particularly high risk for cardiovascular, cerebrovascular, and peripheral artery disease.
Diabetes is characterized by recurrent or persistent hyperglycemia (high blood glucose), and is diagnosed by demonstrating any one of the following:[13]
- Fasting blood glucose level = 126 mg/dl (= 7.0 mmol/L)
- 2 hours glucose tolerance test: Plasma glucose = 200 mg/dL (= 11.1 mmol/L)
- HbA1C IFCC = 48 mmol/mol (6.5%)
A positive result should be confirmed by a repeat on a different day. People with fasting glucose levels from 110 to 125 mg/dl (6.1 to 7.0 mmol/L) are considered to have impaired fasting glucose.[14] People with blood glucose at 140 mg/dL ~ 200 mg/dL (7.8 ~ 11.1 mmol/L) as glucose tolerance test result are considered to have impaired glucose tolerance, which in particular is a major risk for progression to diabetes, as well as cardiovascular disease.[15] HbA1C (glycated hemoglobin), which is an indicator of average blood glucose level, is better than fasting glucose for determining risks of cardiovascular disease and death from any cause.[16] Measuring HbA1C assesses the effectiveness of therapy by monitoring long-term serum glucose regulation, and patients with diabetes who manage to keep their HbA1C level below 53 mmol/mol (7%) are considered to have good glycemic control.
Epidemiology and cardiovascular risk of diabetes
The age-specific prevalence of diabetes rises with age up to the seventh to eighth decades in both men and women.[17] The prevalence is less than 10% in subjects below the age of 60, and 10–20% between 60 ~ 69 years; 15–20% in the oldest groups have previously known diabetes; and a similar proportion have screen-detected asymptomatic diabetes. This suggests that the lifetime risk of diabetes in European people is 30–40%. The prevalence of impaired glucose tolerance increases linearly with age, but the prevalence of impaired fasting glycaemia does not. In middle aged people, the prevalence of impaired glucose homeostasis is about 15%, whereas it is 35–40% in the elderly. The prevalence of diabetes and impaired glucose tolerance defined by isolated post-load hyperglycemia is higher in women than in men, but the prevalence of diabetes and impaired fasting glucose diagnosed by isolated fasting hyperglycemia is higher in men than in women.
Several prospective studies have unequivocally confirmed that post-load hyperglycemia increases cardiovascular diseases morbidity and mortality, however, it remains to be demonstrated that lowering a high 2-hours post-load blood glucose will reduce the risk. Studies are underway but thus far data are scarce. A secondary endpoint analysis of the STOP-NIDDM (Study to Prevent Non-Insulin-Dependent Diabetes Mellitus) revealed statistically significant reductions in cardiovascular diseases event rates in impaired glucose tolerance subjects receiving acarbose compared with placebo.[6] Since acarbose specifically reduces post-prandial glucose excursions, this is the first demonstration that lowering post-prandial glucose and may lead to a reduction in cardiovascular diseases events, but it should be noted that the power in this analysis is low due to a small number of cases.
Identification of subjects at high risk for CVD
Predicting risk of cardiovascular disease in diabetics can be done with the risk score of the DECODE study. The Diabetes Epidemiology: Collaborative Analysis Of Diagnostic Criteria in Europe (DECODE) group developed a cardiovascular diseases risk score, which is presently the only one of its kind including impaired glucose tolerance or impaired fasting glucose in the risk function determination.[7] The large European DECODE study is a collaborative prospective study of 22 cohorts in Europe with baseline glucose measurements for 29714 subjects aged 30-89 years who were followed-up for 11 years, indicated that either fasting or 2-hours post-load blood glucose is an independent risk factor for all-cause and cardiovascular morbidity and mortality even in people without diagnosed diabetes.[18]
Predicting risk to develop diabetes is possible with the Finnish Diabetes Risk Score.[19] The Finnish Diabetes Risk Score predicts the 10 year risk for developing type 2 diabetes with 85% accuracy. It also detects asymptomatic diabetes and abnormal glucose tolerance with high reliability in other populations, which can be used as a self-administered test to screen subjects at high risk for type 2 diabetes, and can also be used in the general population and clinical practice to identify undetected type 2 diabetes, and the abnormal glucose tolerance. In addition, Finnish Diabetes Risk Score is a good predictor that can be used to predict coronary artery disease, stroke and total mortality.[20] Such a simple scoring system can be used to identify high-risk individuals. If under proper management, it can be applied to not only diabetes prevention, but also to cardiovascular diseases prevention.
Treatments to reduce cardiovascular risk
| Treatment to reduce cardiovascular risk [21] | ||
|---|---|---|
| Lifestyle and comprehensive management | ||
| Recommendation | Classa | Levelb |
| Structured patient education improves metabolic and blood pressure control | I | A |
| Non-pharmacological life style therapy improves metabolic control | I | A |
| Self-monitoring improves glycaemic control | I | A |
| Near normoglycaemic control (HbA1c ≤ 6.5%c)
reduces microvascular complications reduces macrovascular complications |
I | A |
| Intensified insulin therapy in type 1 diabetes reduces morbidity and mortality | I | A |
| Early escalation of therapy towards predefined treatment targets improves a composite of morbidity and mortality in type 2 diabetes | IIa | B |
| Early initiation of insulin should be considered in patients with type 2 diabetes failing glucose target | IIb | C |
| Metformin is recommended as first line drug in overweight type 2 diabetes | IIa | B |
| aClass of recommendation.
bLevel of evidence. cDiabetes Control and Complication Trial-standardized. | ||
To reduce the risk of cardiovascular disease, the followings are needed to be considered:
a) the prevention of the progression of diabetes;
b) the prevention of cardiovascular disease by physical activity; and
c) the treatments to reduce cardiovascular risk.
With regarding the first treatment – the prevention of the progression of diabetes, clinical studies have demonstrated that effective lifestyle intervention strategies and drug treatments can prevent or at least delay the progression to type 2 diabetes in high-risk individuals. For instance, the Finnish Diabetes Prevention Study found that a 5% reduction in bodyweight, achieved through an intensive diet and exercise program was associated with a 58% reduction in the risk of developing type 2 diabetes in overweight subjects with impaired glucose tolerance;[22] and the US Diabetes Prevention Program found that lifestyle modification reduced the incidence of type 2 diabetes by 58% in overweight adults with impaired glucose tolerance.[23]
As the second treatment, the prevention of cardiovascular disease by physical activity, The International Diabetes Federation (European Region) have recommended physical activity for the prevention of cardiovascular disease complications among diabetic patients,[24] since studies found that physical activity was associated with reduced risk of cardiovascular disease, cardiovascular death, and total mortality in men with type 2 diabetes. People physically active at their work had a 40% lower cardiovascular mortality compared with people with lower physical activity at work. A high level of leisure-time physical activity like walking and walking pace was associated with a 33% drop in cardiovascular mortality, and moderate physical activity was linked to a 17% drop in cardiovascular mortality compared with the most sedentary group.[25]
To reduce cardiovascular risk in both type 1 and type 2 diabetes long-term hyperglycemia should be treated aggressively. These patients often have a combination of the major vascular risk factors known as the metablolic syndrome.[26][27] The additional treatment of metabolic syndrome is strongly based on non-pharmacological therapy including lifestyle changes, self-monitoring, and requires structured patient education including a heavy emphasis on smoking cessation, etc.[28][29][30] Specific recommendations include 30 min of physical activity for at least five times weekly, increase of fiber uptake to 30g daily, restriction of calorie intake to 1500 calorie daily, restriction of fat intake to 30–35% of total daily uptake, avoidance of trans-fats, and avoidance of liquid mono- and disaccharides. And the most importance of them, a tight glycemic control which will be described in the CAD treatment section.
CAD and diabetes
Case Report
Max is a 53 year old male administrator who had had high blood glucose for 12 months, and a random reading exceeds 342 mg/dL (18.9 mmol/L), blood glucose HBA1C 83.6 mmol/L (9.8%), normal blood pressure 136/92 mmHg on an office visit with a body mass index of 29. He started feeling a little tired recently and was getting up at night to urinate two to three times weekly. Max was eating as usual with no diabetes meal plan at this time, and had limited activities and rare exercise monthly. A few months ago, he got a myocardial infarction and was taking some cardiovascular and hypertensive medications. Max was trying to communicate with his physician and to search the internet to find out some effective solutions to reduce his risks of progression of his coronary artery disease and other cardiovascular complications.
Introduction
The most common cause of death in European diabetic adults is coronary artery disease (CAD). Studies have demonstrated that the risk is two to three times higher than that among people without diabetes.[31] The prevalence of coronary artery disease in patients with type 1 or 2 diabetes are widely different.[32][33] In the EURODIAB IDDM Complication Study which involved 3250 type 1 diabetic patients from 16 countries, the prevalence of cardiovascular disease was 9% in men and 10% in women; and it is increasing with age that it is 6% in the age group of 15–29 years and 25% in the age group of 45–59 years. In men, duration of diabetes was longer, waist-to-hip ratio greater, and hypertension more common in patients with cardiovascular disease, while in women, a greater body mass index was associated with increased prevalence of cardiovascular disease. The risk of coronary artery disease in type 1 diabetic patients increases dramatically when they have the onset of diabetic nephropathy.
Diabetic men and women had comparable mortality rates, whereas coronary mortality among men was significantly higher.[35] Further evidences of the important relations between diabetes and myocardial infarction were obtained from the INTERHEART study in Canada. Diabetes increased the risk of myocardial infarction by more than two times in men and women, and independent of ethnicity. And thus a history of diabetes and myocardial infarction increased cardiovascular disease and mortality markedly. Diabetes or hyperglycemia itself and its complications are very important for the increased risk for coronary artery disease and related mortality.
Evaluation
Diabetes is commonly considered as a coronary artery disease risk equivalent. High-risk factors for coronary artery disease include: [21][36][37]
Typical or atypical symptoms
- 55 years of age or older
- Peripheral or carotid vascular disease
- And plus 2 or more of the following factors: hyperlipidemia, hypertension, smoking, family history of premature coronary artery disease, microalbuminuria, and progressive retinopathy.
Coronary artery disease is associated with smoking, diabetes, and hypertension. Limitation of blood flow to the heart by the artery may cause angina (chest pain) that occurs regularly with activity, after heavy meals, or at other predictable times due to microvascular dysfunction. Detection of coronary artery disease involves the usual diagnostic methods which include baseline electrocardiography (ECG), exercise stress testing ischemic ECG, myocardial perfusion scintigraphy, stress echocardiography, exercise radioisotope test (myocardial scintigraphy), coronary angiography, intravascular ultrasound and magnetic resonance imaging (MRI).
Treatments and outcomes
| Treatment options based on accumulated evidence [21] |
|---|
|
The preventive modalities include doctors counsel exercise, the “diabetes with CAD meal plan” aiming at long-term weight loss, aspirin in doses of 75 to 81 mg/d,[39] (in patients who do not tolerate or have a contra-indication to aspirin, clopidogrel can be used as an alternative antiplatelet agent [40]), antihypertensive therapy and glycemic control.
The optimization of glycemic control is important for prevention and control of any diabetes related cardiovascular diseases.[10] In addition to controlling cardiovascular risk factors, patients with diabetes should aim for good glycemic control (HbA1C < 53 mmol/mol or < 7% for all patients and, for the individual patient, an HbA1C as close as to normal (< 42 mmol/mol or < 6%) as possible) soon after the diagnosis of diabetes to prevent macrovascular as well as microvascular complications. Glycemic targets should be individualized according to the diabetes progression, comorbidities development, and the avoidance of the side effects of therapy (hypoglycemia and weight gain).
There is no simple and effective treatment for coronary artery disease currently. Therapeutic options are based on three principles:[41] 1) Medical treatment – medications like cholesterol lowering medications, beta-blockers, nitroglycerin, calcium antagonists, etc.; 2) Coronary interventions as angioplasty and coronary stent-implantation; 3) Coronary artery bypass grafting (CABG - coronary artery bypass surgery). Recent research progress focuses on new angiogenic treatment modalities (angiogenesis) and various stem cell therapies.
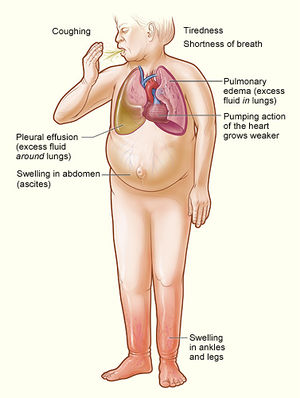
Heart failure and diabetes
Case Report
The 56-year-old plumber Rick was taking medications from his physician to protect his heart and brain, and he felt fluid retention, breathlessness and fatigue most recently. His clinical checkup data were: random blood glucose 388 mg/dL (21.5 mmol/L), fasting glucose 334 mg/dL (18.5 mmol/L), HbA1C 121.8 mmol/L (13.3%, his HbA1C had steadily increased over the past 8 months), total cholesterol 147 mg/dL (8.2mmol/L), HDL 78 mg/dL (4.3 mmol/L), LDL 82 mg/dL (4.6 mmol/L), Trigs 75 mg/dL (4.1 mmol/L), blood pressure 168/98. His physician recommended a diet plan but he refused since he had fast food daily due to his job and he loved chocolates.
Three months ago while Rick was on anti-hypertension medications, had a fall when he was in hospital for severe ‘flu’. Further tests in hospital diagnosed that he had a severely reduced left ventricular ejection fraction and diabetic nephropathy.
Introduction
Prevalence of diabetes and heart failure are increasing exponentially worldwide [42]. Diabetes is well-known to increase the risk of heart failure independent of other traditional risk factors and ischemia. Most heart failure in people with diabetes results from coronary artery disease, and diabetic cardiomyopathy is only said to exist if there is no coronary artery disease to explain the heart muscle disorder.[43] Little is known about the prevalence of the combination of diabetes and heart failure. The most recent and extensive data on the prevalence of diabetes and heart failure are from the Reykjavi´k Study, showing that the prevalence of the combination of heart failure and diabetes is 0.5% in men and 0.4% in women, increasing with increasing age. Heart failure was found in 12% of those with diabetes compared with only 3% in individuals without diabetes.[44] Thus, there was a strong association between diabetes and heart failure. In the Framingham study, the incidence of heart failure was double among males and five times higher in females with diabetes during 18 years of follow-up, compared with patients free from diabetes,
Several clinical and experimental studies have shown that diabetes mellitus leads to functional, biochemical, and morphological abnormalities of the heart, independent of promoting myocardial ischemia, and some of these changes happen earlier in the natural history of diabetes.[42] Diabetes is an independent risk factor for heart failure, and promotes myocardial remodeling (a precursor of heart failure), and the mechanisms beyond ischemia that lead to the development of heart failure in individuals with varying degrees of impaired glucose homeostasis. The most common abnormality observed in asymptomatic diabetics is left ventricular diastolic dysfunction, likely resulting from greater left ventricular myocardial and vascular stiffness. There is also growing evidence that some, if not all, of these structural and biochemical myocardial abnormalities start at the pre-diabetic stage.
Evaluation
The diagnosis of heart failure can be difficult, especially in the early stages. Although symptoms bring patients to medical attention, many of the symptoms of heart failure are non-specific and do not, therefore, help discriminate between heart failure and other problems. Symptoms that are more specific (i.e. orthopnea and paroxysmal nocturnal dyspnea) are less common, especially in patients with milder symptoms, and are therefore insensitive.[45] The echocardiogram and ECG are the most useful tests in patients with suspected heart failure. The echocardiogram provides immediate information on chamber volumes, ventricular systolic and diastolic function, wall thickness, and valve function. The ECG shows the heart rhythm and electrical conduction, i.e. whether there is sinoatrial disease, atrio-ventricular block, or abnormal intra-ventricular conduction. The ECG may also show evidence of left ventricular hypertrophy or Q waves (indicating loss of viable myocardium), giving a possible clue to the etiology of heart failure.
Routine biochemical and hematological investigations are also important to determine whether renin–angiotensin– aldosterone blockade can be initiated safely for renal function and potassium, to exclude anemia which can mimic or aggravate heart failure, and to provide other, useful information. Natriuretic peptide levels and Chest X-ray can be applied as references.
Treatments and outcomes
| Heart failure and diabetes[21] | ||
|---|---|---|
| Recommendation | Classa | Levelb |
| ACE-inhibitors are recommended as first-line therapy in diabetic patients with reduced left ventricular dysfunction with or without symptoms of heart failure | I | C |
| Angiotensin-II receptor blockers have similar effects in heart failure as ACE-inhibitors and can be used as an alternative or even as added treatment to ACE-inhibitors | I | C |
| Beta blockers (metoprolol, bisoprolol, and carvedilol) are recommended as first-line therapy in diabetic patients with heart failure | I | C |
| Diuretics, in particular loop diuretics, are important for symptomatic treatment of diabetic patients with fluid overload owing to heart failure | IIa | C |
| Aldosterone antagonists may be added to ACE-inhibitors, BBs, and diuretics in diabetic patients with severe heart failure | IIb | C |
| aClass of recommendation.
bLevel of evidence. | ||
According to the European Society of Cardiology (ESC) and the European Association for the Study of Diabetes (EASD) Guidelines,[32] there are very few clinical trials on heart failure treatment specifically for diabetic patients. Information on treatment efficacy of various drugs is therefore based on diabetic subgroups included in various heart failure trials. A disadvantage of this is that the subgroups are not always well defined as regards the diabetic state and treatment. Most data favor a proportionately similar efficacy in patients with and without diabetes. Traditional treatment of heart failure in diabetic patients is based on diuretics, ACE-inhibitors, and Beta-blockades, as outlined in other guidelines.[46] Moreover, it is assumed that meticulous metabolic control should be beneficial in heart failure patients with diabetes.
Diuretics are mandatory for relief of symptoms that are due to fluid overload. These drugs should, however, not be used in excess since they induce neuro-hormonal activation.[46] ACE-inhibitors are beneficial in moderate-to-severe heart failure with and without diabetes, they inhibit angiotensin-converting enzyme, thereby decreasing the tension of blood vessels and blood volume, thus lowering blood pressure. Frequently prescribed ACE inhibitors include perindopril, captopril, enalapril, lisinopril, and ramipril. Hypoglycaemia has been reported following the institution of ACE-inhibitors in patients with diabetes on glucose-lowering treatment.[47] It is therefore recommended to monitor blood glucose carefully in the early phase of the institution of an ACE-inhibitor in such patients. Beta-blockade decreases myocardial free fatty acid exposure, thereby changing that metabolic pathway in type 2 diabetes.[48] The addition of eplerenone, a selective aldosterone blocker, to optimal medical therapy reduces morbidity and mortality among patients with acute myocardial infarction complicated by left ventricular dysfunction and heart failure.[49]
Atrial fibrillation and diabetes
Introduction
Evidences show that, apart from coronary artery disease, diabetic patients are at increased risk of arrhythmias.[50] The underlying risk factors for an arrhythmogenic substrate in patients with diabetes include imbalance in autonomic tone, silent ischemia, slowed conduction, heterogeneities in atrial and ventricular repolarization, and the extent of myocardial damage and scar formation.
Atrial fibrillation is relatively common in type 2 diabetes and is associated with substantially increased risks of death and cardiovascular events in patients with type 2 diabetes.[50] This arrhythmia identifies individuals who are likely to obtain greater absolute benefits from blood pressure-lowering treatment. Atrial fibrillation in diabetic patients should be regarded as a marker of particularly adverse outcome and prompt aggressive management of all risk factors. Atrial fibrillation is commonly observed in diabetic patients with prevalence rates estimated to be at least double than those among people without diabetes, and up to three times higher in patients with coexistent hypertension.[51]
Treatments and outcomes
| Arrhythmias: atrial fibrillation and sudden cardiac death[21] | ||
|---|---|---|
| Recommendation | Classa | Levelb |
| Aspirin and anticoagulant use as recommended for patients with atrial fibrillation should be rigorously applied in diabetic patients with atrial fibrillation to prevent stroke | I | C |
| Chronic oral anticoagulant therapy in a dose adjusted to achieve a target international normalized ratio (INR) of 2–3 should be considered in all patients with atrial fibrillation and diabetes, unless contraindicated | IIa | C |
| Control of glycaemia even in the pre-diabetic stage is important to prevent the development of the alterations that predispose to sudden cardiac death | I | C |
| Microvascular disease and nephropathy are indicators of increased risk of sudden cardiac death in diabetic patients | IIa | B |
|
aClass of recommendation. bLevel of evidence. | ||
Aspirin and anticoagulant use as recommended for patients with atrial fibrillation should be rigorously applied in diabetic patients with atrial fibrillation to prevent heart stroke. Oral anticoagulation is most beneficial for patients at higher risk for stroke, whereas the risks outweigh the benefit in patients at low risk.[52] Thus, quantifying the risk of stroke is crucial for determining which atrial fibrillation patients would benefit most from anticoagulant therapy.
Based on the CHADSVASC risk score, all patient with diabetes and atrial fibrillation have an indication for anti-coagulation.[53]
Diabetic patients have a higher incidence of cardiac arrhythmias, including ventricular fibrillation and sudden death. The causes underlying the increased vulnerability of the electrical substrate in these patients are unclear and it is likely to be the consequence of the interplay of several concomitant factors. (i) Atherosclerosis [54] and (ii) microvascular disease [55] are increased in patients with diabetes and they concur to the development of ischemia that pre-disposes to cardiac arrhythmias. (iii) Diabetic autonomic neuropathy [56][57] leads to abnormal reflexes and innervation of the diabetic heart influencing electrical instability. (iv) The electrocardiogram of diabetic patients presents repolarization abnormalities manifesting as prolonged QT interval and altered T waves [57] that may reflect abnormal potassium currents.[58]
Microvascular disease and nephropathy are indicators of increased risk of sudden cardiac death in diabetic patients and should be under well controlled.
Control of glycaemia even in the pre-diabetic stage is important to prevent the development of the alterations that pre-dispose to sudden cardiac death. Microvascular disease and nephropathy are indications of increased risk of sudden cardiac death in diabetic patients.
Peripheral and cerebrovascular diseases and diabetes
Case Report
Simone is a 72-year old lady. She could not remember when she was diagnosed of type 2 diabetes. In one afternoon, while she was eating her late lunch with her husband, he noticed that she was no longer answering his questions and her lower jaw is slightly misplaced. Her husband tried to put on some cold packs to somehow soften the hardened jaw part but it didn’t work. So he drove her to the local hospital for check-up, but her doctor ordered for admission immediately.
As the consequence, Simone was diagnosed with an embolic stroke and atrial fibrillation at the time of admission. A few days after admission, Simone was getting worse. She could hardly speak and open her eyes, and was unable to swallow fluids. Her doctor had to order nasogastric tube insertion and oxygen administration. Her vital signs were: body temperature of 37.2 degree Celsius, blood pressure of 142/94 mmHg, pulse rate of 84 beats per minute of irregular rhythm, and respiratory rate of 24 cycles per minute. She looked generally weak and did not respond to questions, and not even to painful stimuli, and she elicited rates and crackling sounds during respiration, and was diagnosed of type 2 diabetes with emblic stroke with hemorrhagic conversion.
Introduction
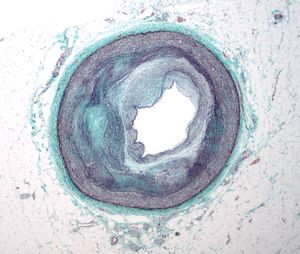
Diabetes related peripheral and cerebrovascular diseases are peripheral vascular disease and stroke. Peripheral vascular disease (PVD, also known as peripheral artery disease, PAD) is a term used to refer to atherosclerotic blockages found in the lower extremity, and it causes either acute or chronic ischemia (lack of blood supply). It has a two- to four-fold increased incidence in subjects with diabetes, which is present in 15% of diabetic patients. The ankle/brachial systolic blood pressure index, a noninvasive measure of peripheral vascular disease, is widely used in epidemiological studies.[59][60] The symptomatic manifestations of peripheral vascular disease are intermittent claudication and limb ischemia. Impairment of the circulation in the foot owing to diabetic macro- and microvascular diseases is the most common non-traumatic reason for limb amputation. The prevalence of peripheral vascular disease increases with advancing age, duration of diabetes, and peripheral neuropathy. The latter condition may mask the symptoms of limb ischemia and thus disease progression may be advanced before patients and healthcare providers realize that peripheral vascular disease is present. About 20% of patients with mild peripheral vascular disease may be asymptomatic; other symptoms include:[61]
- Claudication like pain, weakness, numbness, or cramping in muscles
- Sores, wounds, or ulcers that heal slowly or not at all
- Noticeable change in color or temperature when compared to the other limb, or to both limbs
- Diminished hair and nail growth on affected limb and digits.
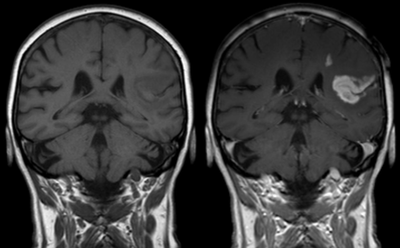
Cerebrovascular disease is a group of brain dysfunctions related to problematic blood vessels supplying the brain, and hypertension is the most important cause.[62] The typical cerebrovascular disease in diabetes is brain stroke, which is the second leading cause of death worldwide. A transient ischemic attack (minor stroke) leaves little to no permanent damage in the brain, which symptoms include facial weakness, visual impairment, loss of coordination or balance, sudden headache, and mental confusion with unintelligible speech. And symptoms of a cerebral contralateral (opposite sided) include paralysis of a single body part; paralysis of one side of the body; localized tingling, numbness; hemianopia visual loss; aphasia (loss of speech); even loss of consciousness.
Cerebrovascular mortality rates have been shown to be raised in patients with type 2 diabetes and have been reported that it is at least as great a risk factor in type 1 diabetes as in type 2. It is found that cerebrovascular mortality is raised at all ages in these patients.[63] Typical brain strokes can be classified into two categories: ischemic and hemorrhagic strokes.[62] Ischemic strokes are those that are caused by vessel occlusion mostly by embolism, while hemorrhagic strokes (intracerebral hemorrhage) are the ones which result from rupture of a blood vessel due to hypertension. About 80% of strokes are caused by ischemia, and the remainder by hemorrhage. Some hemorrhages develop inside the areas of ischemia, and thus it is unknown how many hemorrhages actually start as ischemic stroke.
Evaluation
Peripheral vascular diseases: An objective measure of peripheral vascular disease is the ankle–brachial blood pressure index, defined as the ratio between the arterial pressure at the ankle level and in the brachial artery with the highest pressure,[59] which means that when the blood pressure reading in the ankles is lower than that in the arms, blockages in the arteries providing blood from the heart to the ankle are suspected. The ankle–brachial blood pressure index should normally be 0.9. This measurement is valuable for early detection of peripheral artery disease and also for a better stratification of overall cardiovascular risk. A 0.5 or an ankle pressure as 50 mm Hg indicates severely impaired circulation to the foot, below 0.8 indicates moderate ischemic disease, and below 0.5 implies severe ischemic disease, alternatively a 0.4 is used as a threshold. An index above 1.3 indicates poorly compressible vessels as a result of stiff arterial walls, which in diabetic patients are due to atherosclerosis in the media layer of the arterial wall.
Further traditional examinations include a lower limb Doppler ultrasound, an angiography where a catheter is inserted into the common femoral artery and selectively guided to the artery, or an X-ray by injecting a radio dense contrast agent to observe the artery directly. On the other hand, modern and non-invasive technologies like multislice computerized tomography (CT) scanners and magnetic resonance imaging (MRI) provide direct imaging of the aorta and lower limb arteries without the need for an injection of contrast agents and as an alternative to angiography.
Cerebrovascular diseases: After transient ischemic attack or minor stroke, the risk of further stroke is substantially higher than previously thought, reaching as high as 30% within the first month in some subgroups.[62] Patients at very high risk (>30%) of recurrence within 7 days can be identified on the basis of their ages, blood sugar levels, blood pressures, and other symptoms. Simple risk scores have been developed on the basis of these factors to predict those patients at greatest risk who might benefit the most from early risk-factor modifications.
Both the ischemic and hemorrhagic strokes are diagnosed through the following techniques: a neurological body examination, Doppler ultrasound, arteriography, and CT and MRI scans of the brain. The imaging techniques will assist in determining the subtypes and causes of stroke. And blood tests may be helpful in finding out the causes of stroke.
Treatments and outcomes
| Peripheral vascular disease[21] | ||
|---|---|---|
| Recommendation | Classa | Levelb |
| All patients with type 2 diabetes and CVD are recommended treatment with low-dose aspirin | IIa | B |
| In diabetic patients with peripheral vascular disease, treatment with clopidogrel or low molecular weight heparin may be considered in certain cases | IIa | B |
| Patients with critical limb ischaemia should, if possible, undergo revascularization procedures | I | B |
| An alternative treatment for patients with critical limb ischaemia, not suited for revascularization, is prostacyclin infusion | I | A |
|
aClass of recommendation. bLevel of evidence. | ||
Peripheral vascular diseases: Preventions are most important strategies, such as regular exercises, the optimization of glycemic control, management of hypertension, smoking cessation, antiplatelet and anti-cholesterol medications like aspirin, clopidogrel and statins.
For diabetic patients with peripheral vascular disease, general measures to reduce overall cardiovascular risk should be intensive, and treatment of hypertension should be vigorous. But in patients with critical limb ischemia and very low distal perfusion pressures, it may be dangerous to lower blood pressure too much for the foot, and thus blood pressure should be kept at a level permitting adequate arterial inflow to the distal extremity. Platelet inhibition with low-dose aspirin, in the magnitude of 75–250 mg per day, is indicated in all patients with severe peripheral vascular disease; further inhibition of platelet aggregation by clopidogrel or dipyridamole along with anticoagulation with low molecular weight heparin may be the first set of choice.[64][65][66]
If anatomically possible, a revascularization procedure should be attempted in all patients with critical limb ischemia,[67] such as an angioplasty or a bypass grafting can be done on solitary lesions in large arteries like the femoral artery, but the revascularization may not have sustained benefits. A synthetic prostacyclin (Ilomedin, Iloprost) is the only pharmacological agent so far convincingly shown to have significant beneficial effects on ulcer healing and pain relief on patients with critical limb ischemia, which is given intravenously daily for a period of 2–4 weeks.[68]
| Stroke[21] | ||
|---|---|---|
| Recommendation | Classa | Levelb |
| For stroke prevention, blood pressure lowering is more important than the choice of drug. Inhibition of the renin–angiotensin–aldosterone system may have additional benefits beyond blood pressure lowering per se | IIa | B |
| Patients with acute stroke and diabetes should be treated according to the same principles as stroke patients without diabetes | IIa | C |
|
aClass of recommendation. bLevel of evidence. | ||
Cerebrovascular diseases: Stroke prevention should be based on a multifactorial strategy aimed at the treatment of hypertension, hyperlipidemia, microalbuminuria, hyperglycemia and the use of antiplatelet medications. Antiplatelet therapy reduces the incidence of stroke in diabetic patients and is indicated for both primary and secondary prevention.[69] Low dose Aspirin (75–250 mg daily) should be the initial choice, but in case of intolerance, clopidogrel 75 mg once daily should be given.[70] In patients with recurrent stroke, a combination of aspirin and dipyridamol should be a better option.
After a transient ischemic attack or stroke, since complications are more frequent in diabetic subjects compared with non-diabetic subjects, special attention should be paid to the overall risk for peri- and post-operative morbidity and mortality when making decision on surgical interventions, and a simple risk score that can be used to stratify patients into different risk groups for complications after surgery has been developed.[71] An alternative to endoarterectomy is carotid artery angioplasty and carotid artery stenting, which has been found to be not inferior to endoarterectomy and may prove to be a preferable method in high-risk patients.[72]
The treatment in the acute phase of stroke in diabetic patients should follow the same principles as in the general population, and patients should be kept in stroke care units as the routine management to close surveillance of vital functions and optimization of circulation and metabolic conditions.[62]Thrombolysis like recombinant tPA is one of the most effective treatments but requires application within 3–4 h of stroke onset, which reduces mortality and disability from stroke, but is associated with a risk of hemorrhage. Because of the substantial effectiveness of tPA, efforts to increase the number of patients who are eligible for thrombolytic therapy, and combination of tPA with modern imaging technologies are underway.[62] Hemispheric decompression in young patients with malignant middle cerebral artery territory infarction and space occupying brain edema is supported by evidence. Mechanical thrombectomy and hemicraniectomy could be other options.[73][74]
As reviewed recently, strict blood glucose control with intensive insulin therapy improved mortality and morbidity of adult diabetic stroke patients. Besides saving lives, intensive insulin therapy largely prevented several critical illness-associated complications including critical illness polyneuropathy, blood stream infections, anemia, and acute renal failure.[75] But arguments of the safety of the intensive insulin therapy on diabetic patients are ongoing, and new evidences show that diabetic critical-ill patients react differently to the therapy compared to non-diabetic ones, and a carbohydrate-restrictive strategy could be safer and as efficient as intensive insulin therapy, and thus could be a replacement in the near future.[76][77]
References
Error fetching PMID 10413697:
Error fetching PMID 17192512:
Error fetching PMID 17353438:
Error fetching PMID 430798:
Error fetching PMID 20609967:
Error fetching PMID 9686693:
Error fetching PMID 16194123:
Error fetching PMID 20200384:
Error fetching PMID 12502659:
Error fetching PMID 12876091:
Error fetching PMID 15662552:
Error fetching PMID 12610023:
Error fetching PMID 16305061:
Error fetching PMID 16210931:
Error fetching PMID 12687329:
Error fetching PMID 11832527:
Error fetching PMID 14632698:
Error fetching PMID 12719277:
Error fetching PMID 11315831:
Error fetching PMID 14706054:
Error fetching PMID 11772915:
Error fetching PMID 11874956:
Error fetching PMID 15793191:
Error fetching PMID 10331395:
Error fetching PMID 11023146:
Error fetching PMID 8799621:
Error fetching PMID 15364185:
Error fetching PMID 17220161:
Error fetching PMID 10749967:
Error fetching PMID 17291928:
Error fetching PMID 17488967:
Error fetching PMID 17506599:
Error fetching PMID 22108732:
Error fetching PMID 15094099:
Error fetching PMID 15735197:
Error fetching PMID 22828712:
Error fetching PMID 15901669:
Error fetching PMID 9283780:
Error fetching PMID 12106931:
Error fetching PMID 12668699:
Error fetching PMID 19282274:
Error fetching PMID 16274775:
Error fetching PMID 10507957:
Error fetching PMID 22922413:
Error fetching PMID 7867189:
Error fetching PMID 9484982:
Error fetching PMID 18468545:
Error fetching PMID 12574553:
Error fetching PMID 8918275:
Error fetching PMID 11372014:
Error fetching PMID 12941721:
Error fetching PMID 10666287:
Error fetching PMID 7525794:
Error fetching PMID 9243111:
Error fetching PMID 8981292:
Error fetching PMID 14526040:
Error fetching PMID 16476626:
Error fetching PMID 22182267:
Error fetching PMID 21190097:
Error fetching PMID 19531590:
Error fetching PMID 21871056:
Error fetching PMID 19327317:
Error fetching PMID 16399854:
Error fetching PMID 23996285:
-
Shoback, edited by David G. Gardner, Dolores (2011). Greenspan's basic & clinical endocrinology (9th ed.). New York: McGraw-Hill Medical. pp. Chapter 17. ISBN 0-07-162243-8
- Error fetching PMID 10413697:
- Error fetching PMID 17192512:
- Error fetching PMID 117544:
- Error fetching PMID 12876091:
- Error fetching PMID 15662552:
- Error fetching PMID 17353438:
- Error fetching PMID 430798:
- Error fetching PMID 20609967:
-
DeFronzo RA. International Textbook of Diabetes Mellitus. 3rd ed. Chichester, West Sussex; Hoboken, NJ: John Wiley; 2004.
- Error fetching PMID 9686693:
- Error fetching PMID 16194123:
- Error fetching PMID 20200384:
- Error fetching PMID 12502659:
- Error fetching PMID 12610023:
- Error fetching PMID 16305061:
- Error fetching PMID 16210931:
- Error fetching PMID 17220161:
- Error fetching PMID 12687329:
- Error fetching PMID 11832527:
- Error fetching PMID 14632698:
- Error fetching PMID 12719277:
- Error fetching PMID 11315831:
- Error fetching PMID 14706054:
- Error fetching PMID 11772915:
- Error fetching PMID 11874956:
- Error fetching PMID 15793191:
- Error fetching PMID 10331395:
- Error fetching PMID 11023146:
- Error fetching PMID 8799621:
- Error fetching PMID 16399854:
- Error fetching PMID 15364185:
- Error fetching PMID 10749967:
- Error fetching PMID 17291928:
- Error fetching PMID 23996285:
- Error fetching PMID 17488967:
- Error fetching PMID 17506599:
-
Jameson JN, Kasper DL, Harrison TR, Braunwald E, Fauci AS, Hauser SL, Longo DL. Harrison's Principles of Internal Medicine (16th edition). 2005. New York: McGraw-Hill Medical Publishing.
- Error fetching PMID 22108732:
- Error fetching PMID 15094099:
- Error fetching PMID 15735197:
- Error fetching PMID 22828712:
- Error fetching PMID 15901669:
- Error fetching PMID 9283780:
- Error fetching PMID 12106931:
- Error fetching PMID 12668699:
- Error fetching PMID 19282274:
- Error fetching PMID 16274775:
- Error fetching PMID 10507957:
- Error fetching PMID 22922413:
-
pmid=A
-
pmid=B
-
pmid=C
-
pmid=D
-
pmid=E
-
pmid=F
- Error fetching PMID 7867189:
- Error fetching PMID 9484982:
- Error fetching PMID 18468545:
- Error fetching PMID 12574553:
- Error fetching PMID 8918275:
- Error fetching PMID 11372014:
- Error fetching PMID 12941721:
- Error fetching PMID 10666287:
- Error fetching PMID 7525794:
- Error fetching PMID 9243111:
- Error fetching PMID 8981292:
- Error fetching PMID 14526040:
- Error fetching PMID 16476626:
- Error fetching PMID 22182267:
- Error fetching PMID 21190097:
- Error fetching PMID 19531590:
- Error fetching PMID 21871056:
- Error fetching PMID 19327317: