Infective Endocarditis: Difference between revisions
No edit summary |
No edit summary |
||
| (23 intermediate revisions by 2 users not shown) | |||
| Line 1: | Line 1: | ||
''Author: A. Crystal'' | |||
==Introduction== | ==Introduction== | ||
Infective endocarditis (IE) is an infectious and inflammatory process of endothelial lining of the heart structures and valves. It is most commonly caused by bacterial and fungal infections, although non-infective causes of endocarditis occur, this chapter will concentrate on infective causes. | Infective endocarditis (IE) is an infectious and inflammatory process of endothelial lining of the heart structures and valves. It is most commonly caused by bacterial and fungal infections, although non-infective causes of endocarditis occur, this chapter will concentrate on infective causes. | ||
| Line 6: | Line 7: | ||
==Pathogenesis and Causes== | ==Pathogenesis and Causes== | ||
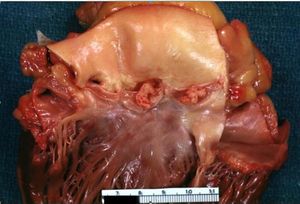
[[Image:Heart1.JPG|thumb|300px|Large lesions on non coronary and left coronary cusps normal valves otherwise]] | |||
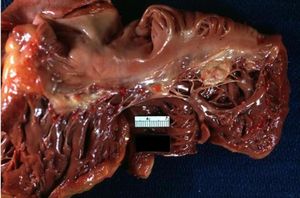
[[Image:Heart2.JPG|thumb|300px|Vegetations on tricuspid valve]] | |||
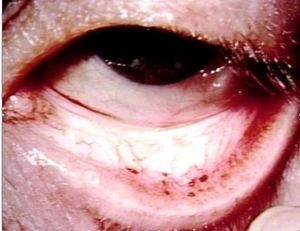
[[Image:Heart3.JPG|thumb|300px|Septic emboli to the conjunctiva]] | |||
Generally, endothelial damage to heart valves predisposes to bacterial infections as it is generally resistant to bacterial infections. This may be caused by turbulent blood flow in damaged valves, septal defects or instrumentation. Although, recent evidence suggests that A-type von Willebrand factor may contribute to S. aureus binding in endothelial intact valves<cite>v</cite>. Specifically, in patients with S. aureus bacteremia, native valve endocarditis was reported to be in 19% of patients, and 38% in those with prosthetic valves<cite>vi</cite>. | Generally, endothelial damage to heart valves predisposes to bacterial infections as it is generally resistant to bacterial infections. This may be caused by turbulent blood flow in damaged valves, septal defects or instrumentation. Although, recent evidence suggests that A-type von Willebrand factor may contribute to S. aureus binding in endothelial intact valves<cite>v</cite>. Specifically, in patients with S. aureus bacteremia, native valve endocarditis was reported to be in 19% of patients, and 38% in those with prosthetic valves<cite>vi</cite>. | ||
While many microorganisms have been implicated in endocarditis syndromes, few infectious bacteria account for the majority of cases. | While many microorganisms have been implicated in endocarditis syndromes, few infectious bacteria account for the majority of cases. | ||
Staphylococcal endocarditis is most commonly caused by S.aureus. It is the most lethal organism implicated in endocarditis with mortality rates approaching 37%<cite>vii</cite>. S.aureus is a highly virulent organism and may cause significant valve destruction, abscess formation, conduction abnormalities and embolization. It often enters the bloodstream from the nares or skin. Patients with left sided involvement often require surgery. In intravenous drug users is the most common cause of IE<cite>viii</cite>. Patients who are found to have Staphylococcal bacteremia should undergo echocardiography to rule out endocarditis. The prevalence of endocarditis in patients with S.aureus bacteremia was reported in 19% and 38% in those with native and prosthetic valves respectively<cite> | Staphylococcal endocarditis is most commonly caused by S.aureus. It is the most lethal organism implicated in endocarditis with mortality rates approaching 37%<cite>vii</cite>. S.aureus is a highly virulent organism and may cause significant valve destruction, abscess formation, conduction abnormalities and embolization. It often enters the bloodstream from the nares or skin. Patients with left sided involvement often require surgery. In intravenous drug users is the most common cause of IE<cite>viii</cite>. Patients who are found to have Staphylococcal bacteremia should undergo echocardiography to rule out endocarditis. The prevalence of endocarditis in patients with S.aureus bacteremia was reported in 19% and 38% in those with native and prosthetic valves respectively<cite>vi</cite>. | ||
S. epidermidis is an important cause of prosthetic valve endocarditis and is associated with a particularly high incidence of heart failure and valvular abscess formation and a mortality rate of 36%<cite> | S. epidermidis is an important cause of prosthetic valve endocarditis and is associated with a particularly high incidence of heart failure and valvular abscess formation and a mortality rate of 36%<cite>ix</cite>. | ||
Viridans group streptococcus often account for 30% of community acquired native valve endocarditis<cite> | Viridans group streptococcus often account for 30% of community acquired native valve endocarditis<cite>x</cite>. They are part of the oral cavity flora and may gain entry into the bloodstream via dental caries or trauma. The virulence is generally low and eradication rates are high. | ||
Streptococcus bovis is part of the lower gastrointestinal and urinary tract and is commonly implicated in underlying colorectal disease if found to be the cause of endocarditis. Patients who are found to have S.bovis endocarditis should undergo a colonoscopy to exclude colorectal malignancy or polyps. | Streptococcus bovis is part of the lower gastrointestinal and urinary tract and is commonly implicated in underlying colorectal disease if found to be the cause of endocarditis. Patients who are found to have S.bovis endocarditis should undergo a colonoscopy to exclude colorectal malignancy or polyps. | ||
| Line 20: | Line 24: | ||
Enterococcal endocarditis is part of the gastrointestinal and genitourinary flora and is often implicated in patients in patients undergoing genitourinary or gastrointestinal procedures. Enterococcal endocarditis is generally difficult to treat due to high rates of antibiotic resistance and often require multi-drug regimen. | Enterococcal endocarditis is part of the gastrointestinal and genitourinary flora and is often implicated in patients in patients undergoing genitourinary or gastrointestinal procedures. Enterococcal endocarditis is generally difficult to treat due to high rates of antibiotic resistance and often require multi-drug regimen. | ||
Gram negative bacilli IE is rather uncommon, the HACEK organisms (Haemophillus spp, Actinobacillus, Cardiobacterium hominis, Eikenella corrodens, Kingella spp) are responsible for approximately 3% of endocarditis cases and are the most common cause for gram negative endocarditis in non-intravenous drug users<cite> | Gram negative bacilli IE is rather uncommon, the HACEK organisms (Haemophillus spp, Actinobacillus, Cardiobacterium hominis, Eikenella corrodens, Kingella spp) are responsible for approximately 3% of endocarditis cases and are the most common cause for gram negative endocarditis in non-intravenous drug users<cite>xi</cite>. Non-HACEK organisms are a rare cause for endocarditis and only account for <1-2% of causes. | ||
Fungal endocarditis occurs in patients who receive prolonged parenteral nutrition or antibiotics through intravenous catheters. It has also been described in intravenous drug users. Patients are often immunocompromised. The most common organisms implicated are Candida species, Histoplasma capsulatum, and Aspergillus. Mortality rates associated with fungal endocarditis exceed 80%<cite> | Fungal endocarditis occurs in patients who receive prolonged parenteral nutrition or antibiotics through intravenous catheters. It has also been described in intravenous drug users. Patients are often immunocompromised. The most common organisms implicated are Candida species, Histoplasma capsulatum, and Aspergillus. Mortality rates associated with fungal endocarditis exceed 80%<cite>xii</cite>. | ||
==Diagnosis== | ==Diagnosis== | ||
Several diagnostic criteria have been proposed for the diagnosis of IE. In clinical practice, it is the global clinical picture that leads to decision making in the diagnosis and treatment of endocarditis. The modified DUKE criteria for diagnosis is often widely used, with a sensitivity and specificity approaching ~80%.<cite> | Several diagnostic criteria have been proposed for the diagnosis of IE. In clinical practice, it is the global clinical picture that leads to decision making in the diagnosis and treatment of endocarditis. The modified DUKE criteria for diagnosis is often widely used, with a sensitivity and specificity approaching ~80%.<cite>xiii</cite> The DUKE criteria divides into Definite IE, Possible IE, or Rejected IE. It uses Major criteria (microbiology, valvular abnormalities) and Minor criteria (systemic symptoms described below). Using the diagnostic criteria for IE should not override clinical judgment. | ||
===Definite IE:=== | ===Definite IE:=== | ||
*Pathologic Criteria: | *Pathologic Criteria: | ||
**Microorganisms demonstrated by culture or histologic examination of vegetation, a vegetation that has embolized, or an intracardiac abscess specimen OR pathologic lesions; vegetation or intracardiac abscess confirmed by histologic examination showing active endocarditis | **Microorganisms demonstrated by culture or histologic examination of vegetation, a vegetation that has embolized, or an intracardiac abscess specimen OR pathologic lesions; vegetation or intracardiac abscess confirmed by histologic examination showing active endocarditis | ||
| Line 50: | Line 38: | ||
===Possible IE:=== | ===Possible IE:=== | ||
*1 Major criterion + 1 Minor criterion '''OR''' 3 minor criteria | *1 Major criterion + 1 Minor criterion '''OR''' 3 minor criteria | ||
===Rejected IE:=== | ===Rejected IE:=== | ||
*Firm alternate diagnosis explaining evidence of IE '''OR''' | *Firm alternate diagnosis explaining evidence of IE '''OR''' | ||
*Resolution of IE syndrome with =<4 days of antibiotics therapy '''OR''' | *Resolution of IE syndrome with =<4 days of antibiotics therapy '''OR''' | ||
| Line 101: | Line 87: | ||
|} | |} | ||
The sensitivity of detecting on echocardiogram varies. Transthoracic and transesophageal echocardiogram sensitivities for detecting vegetations are 50% and 90% respectively.<cite> | The sensitivity of detecting on echocardiogram varies. Transthoracic and transesophageal echocardiogram sensitivities for detecting vegetations are 50% and 90% respectively.<cite>xiv</cite><cite>xv</cite> | ||
{| class="wikitable" cellpadding="0" cellspacing="0" border="0" width=" | {| class="wikitable" cellpadding="0" cellspacing="0" border="0" width="500px" | ||
|- | |- | ||
!colspan="2"|Endocarditis of prosthetic mitral valve | !colspan="2"|Endocarditis of prosthetic mitral valve | ||
|- | |- | ||
|http://www.echopedia.org/images/2/24/E00404.swf | |[http://www.echopedia.org/images/2/24/E00404.swf PLAX: vegetations on PMVL]<br>Video courtesy: AMC Echolab, AMC, The Netherlands | ||
|http://www.echopedia.org/images/6/6a/E00405.swf | |[http://www.echopedia.org/images/6/6a/E00405.swf A4CH]<br>Video courtesy: AMC Echolab, AMC, The Netherlands | ||
|- | |- | ||
!colspan="2"|Endocarditis of the aortic valve | !colspan="2"|Endocarditis of the aortic valve | ||
|- | |- | ||
|http://www.echopedia.org/images/1/10/E00114.swf | |[http://www.echopedia.org/images/1/10/E00114.swf PLAX: showing an aortic valve vegetation]<br>Video courtesy: J. Vleugels, AMC, The Netherlands | ||
|http://www.echopedia.org/images/5/5b/E00117.swf | |[http://www.echopedia.org/images/5/5b/E00117.swf PLAX: aortic valve vegetation]<br>Video courtesy: J. Vleugels, AMC, The Netherlands | ||
|} | |} | ||
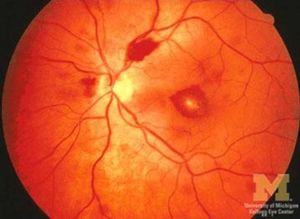
[[Image:Heart4.JPG|thumb|300px|Roth Spots]] | |||
==Therapy== | ==Therapy== | ||
| Line 130: | Line 111: | ||
As soon as blood cultures become available antibiotics should be adjusted to target the identified microorganisms. | As soon as blood cultures become available antibiotics should be adjusted to target the identified microorganisms. | ||
In cases of prosthetic valve endocarditis (PVE), microbiological activity depends on early (<2 months post op) or late (>2 months post op). In early PVE S.aureus accounts for 40% of the cases, followed by coagulase negative staphylococcus (17%). In late PVE coagulase negative staphylococcus accounts for 20% of cases, followed by S. aureus (18%). Coverage for enterococci, streptococci, and gram negative should be considered in empiric therapy in both groups. Rifampin + Vancomycin + Gentamicin should be initiated for PVE <12 post op. Suspected PVE >12 months post op may be treated with the same regimen as for native valves.<cite> | In cases of prosthetic valve endocarditis (PVE), microbiological activity depends on early (<2 months post op) or late (>2 months post op). In early PVE S.aureus accounts for 40% of the cases, followed by coagulase negative staphylococcus (17%). In late PVE coagulase negative staphylococcus accounts for 20% of cases, followed by S. aureus (18%). Coverage for enterococci, streptococci, and gram negative should be considered in empiric therapy in both groups. Rifampin + Vancomycin + Gentamicin should be initiated for PVE <12 post op. Suspected PVE >12 months post op may be treated with the same regimen as for native valves.<cite>xvi</cite> | ||
The American Heart Association recommendation for specific antimicrobial therapy | The American Heart Association recommendation for specific antimicrobial therapy can be found in their [http://circ.ahajournals.org/content/111/23/3167.full guideline]. | ||
http://circ.ahajournals.org/content/111/23/3167.full | |||
The | The European Society of Cardiology [http://eurheartj.oxfordjournals.org/content/30/19/2369.long guideline for the treatment of Infective endocarditis.] | ||
http://eurheartj.oxfordjournals.org/content/30/19/2369.long | |||
===Prophylaxis=== | ===Prophylaxis=== | ||
According the American Heart Association guidelines published in 2007 the following groups of patients are considered to be high-risk and require prophylaxis:<cite>xvii</cite> | |||
According the American Heart Association guidelines published in 2007 the following groups of patients are considered to be high-risk and require prophylaxis:<cite> | |||
*Any prosthetic heart valve, or prosthetic material used for valve repair | *Any prosthetic heart valve, or prosthetic material used for valve repair | ||
| Line 154: | Line 132: | ||
**Incision or biopsy of the respiratory mucosa, or procedures involving treatment of abscess of empyema | **Incision or biopsy of the respiratory mucosa, or procedures involving treatment of abscess of empyema | ||
Antibiotic prophylaxis is also recommended for any procedures on infected skin/skin structures or musculoskeletal tissue in high risk patients.<cite> | Antibiotic prophylaxis is also recommended for any procedures on infected skin/skin structures or musculoskeletal tissue in high risk patients.<cite>xvii</cite> | ||
==Complications of Endocarditis== | ==Complications of Endocarditis== | ||
Common complications arising from IE can be divided into local and systemic. Local complications often arise from direct extension of the infection into cardiac structures. Systemic complications arise from embolization and bacteremias. | Common complications arising from IE can be divided into local and systemic. Local complications often arise from direct extension of the infection into cardiac structures. Systemic complications arise from embolization and bacteremias. | ||
Heart failure occurs in 26-30% of patients with endocarditis.<cite> | Heart failure occurs in 26-30% of patients with endocarditis.<cite>xviii</cite><cite>xix</cite> It may occur acutely or over time, it is often times due to anatomical disruption from valve vegetations or destruction of nearby tissue. Development of heart failure in the setting of IE is correlated with worse outcomes. Heart failure occurs most commonly with aortic (29%) and mitral valve (20%) infections and less with tricuspid valve (8%).<cite>xii</cite> The overall in hospital mortality rate for patients diagnosed with heart failure approaches 30%.<cite>xx</cite> | ||
Conduction abnormalities, commonly characterized by heart blocks in endocarditis are associated with infection extension, increased risk of embolization and increased mortality. They are reported to be present in 26%-28% of patients.<cite> | Conduction abnormalities, commonly characterized by heart blocks in endocarditis are associated with infection extension, increased risk of embolization and increased mortality. They are reported to be present in 26%-28% of patients.<cite>xxi</cite> | ||
Embolization is a dreaded complication of IE and most commonly affects the spleen, brain, kidneys in cases of left sided endocarditis, and the lung in right sided endocarditis. Studies report a rate of 8.5-25% and are associated with significant mortality risk.<cite> | Embolization is a dreaded complication of IE and most commonly affects the spleen, brain, kidneys in cases of left sided endocarditis, and the lung in right sided endocarditis. Studies report a rate of 8.5-25% and are associated with significant mortality risk.<cite>xxii</cite><cite>xxiii</cite> Vegetation length, especially >10mm, infection with S. aureus, S. bovis are predictive factors for a higher rate of embolization and increased in mortality.<cite>xxiv</cite> Embolization to the brain can result in mycotic aneurysms which can present with a variety of neurologic manifestations depending on the anatomic location and spread of infection in the surrounding area. Up to 30% of patients with evidence of embolization to the brain are reported to be asymptomatic.<cite>xxv</cite> | ||
==Prognosis== | ==Prognosis== | ||
Prognosis of IE is largely dependent on the patient’s comorbid conditions such as diabetes,<cite> | Prognosis of IE is largely dependent on the patient’s comorbid conditions such as diabetes,<cite>xxvi</cite> hemodialysis, congestive heart failure,<cite>xxvii</cite> complications of endocarditis, prosthetic valve and the microorganism identified. Generally the outcome largely depends on the organism involved. According to recent data it, the over 30 day mortality is ~15% and the 1-year mortality is ~34%.<cite>xxviii</cite> Prosthetic valve endocarditis has a significant in hospital mortality of ~24%,<cite>xxix</cite> while native valve endocarditis carries a lower in hospital mortality of 12% if treated early and surgically.<cite>xxx</cite> | ||
==References== | ==References== | ||
<biblio> | <biblio> | ||
#i | #i [http://www.nejm.org/doi/full/10.1056/NEJMcp1206782 "Infective Endocarditis - N Engl J Med 2013." 12 Sep. 2013] | ||
#ii Cabell CH, Fowler VG, Jr, Engemann JJ, et al. Endocarditis in the elderly: Incidence, surgery, and survival in 16,921 patients over 12 years. Circulation.2002;106(19):547. | #ii Cabell CH, Fowler VG, Jr, Engemann JJ, et al. "Endocarditis in the elderly: Incidence, surgery, and survival in 16,921 patients over 12 years." Circulation.2002;106(19):547. | ||
#iii | #iii pmid=12092473 | ||
#iv | #iv pmid=22870738 | ||
#v | #v pmid=23720451 | ||
#vi pmid=21685200 | #vi pmid=21685200 | ||
#vii | #vii pmid=22624837 | ||
#viii pmid=16028160 | #viii pmid=16028160 | ||
#ix | #ix pmid=16767483 | ||
#x | #x pmid=19665088 | ||
#xi | #xi pmid=9046942 | ||
#xii | #xii pmid=15956145 | ||
#xiii | #xiii pmid=10770721 | ||
#xiv | #xiv pmid=15145856 | ||
#xv | #xv pmid=2768712 | ||
#xvi pmid= | #xvi pmid=19713420 | ||
#xvii | #xvii pmid=17446442 | ||
#xviii | #xviii [http://www.ncbi.nlm.nih.gov/genome/sts/sts.cgi?uid=85362 The National Center for Biotechnology Information. "UniSTS." 2006. 20 Nov. 2013] | ||
#xix pmid=3259778 | |||
#xx pmid=22110106 | |||
#xxi pmid=11479467 | |||
#xxii | #xxii pmid=12957420 | ||
#xxiii | #xxiii pmid=23906859 | ||
#xxiv | #xxiv pmid=15983252 | ||
#xxv | #xxv pmid=18491965 | ||
#xxvi | #xxvi pmid=16857604 | ||
#xxvii | #xxvii pmid=23906529 | ||
#xxviii pmid= | #xxviii pmid=23994421 | ||
#xxix pmid= | #xxix pmid=16291003 | ||
#xxx | #xxx pmid=20159831 | ||
</biblio> | </biblio> | ||
Latest revision as of 20:46, 8 December 2013
Author: A. Crystal
Introduction
Infective endocarditis (IE) is an infectious and inflammatory process of endothelial lining of the heart structures and valves. It is most commonly caused by bacterial and fungal infections, although non-infective causes of endocarditis occur, this chapter will concentrate on infective causes.
Epidemiology
The global incidence of endocarditis varies in literature with a wide range of 3 to 9 cases per 100,000 person years[1]. Although it appears that in the elderly population the incidence rate is remarkably higher reaching 20.4 cases per 100,000 person years[2]. This is likely related to increased hospitalization, valve replacement surgeries and intra-cardiac instrumentation in the elderly population. In addition to aging, the prevalence of chronic diseases predisposing to bacteremias such as human immunodeficiency syndrome, end-stage renal disease, organ transplantations and diabetes have also increased. IE is a dreaded complication of intravenous drug use, most commonly affecting the tricuspid valve, with a yearly incidence of 1-5% among chronic users[3]. IE related to implantable rhythm devices remains relatively low but is on the rise due to increased patient population requiring rhythm devices[4].
Pathogenesis and Causes
Generally, endothelial damage to heart valves predisposes to bacterial infections as it is generally resistant to bacterial infections. This may be caused by turbulent blood flow in damaged valves, septal defects or instrumentation. Although, recent evidence suggests that A-type von Willebrand factor may contribute to S. aureus binding in endothelial intact valves[5]. Specifically, in patients with S. aureus bacteremia, native valve endocarditis was reported to be in 19% of patients, and 38% in those with prosthetic valves[6].
While many microorganisms have been implicated in endocarditis syndromes, few infectious bacteria account for the majority of cases.
Staphylococcal endocarditis is most commonly caused by S.aureus. It is the most lethal organism implicated in endocarditis with mortality rates approaching 37%[7]. S.aureus is a highly virulent organism and may cause significant valve destruction, abscess formation, conduction abnormalities and embolization. It often enters the bloodstream from the nares or skin. Patients with left sided involvement often require surgery. In intravenous drug users is the most common cause of IE[8]. Patients who are found to have Staphylococcal bacteremia should undergo echocardiography to rule out endocarditis. The prevalence of endocarditis in patients with S.aureus bacteremia was reported in 19% and 38% in those with native and prosthetic valves respectively[6].
S. epidermidis is an important cause of prosthetic valve endocarditis and is associated with a particularly high incidence of heart failure and valvular abscess formation and a mortality rate of 36%[9].
Viridans group streptococcus often account for 30% of community acquired native valve endocarditis[10]. They are part of the oral cavity flora and may gain entry into the bloodstream via dental caries or trauma. The virulence is generally low and eradication rates are high.
Streptococcus bovis is part of the lower gastrointestinal and urinary tract and is commonly implicated in underlying colorectal disease if found to be the cause of endocarditis. Patients who are found to have S.bovis endocarditis should undergo a colonoscopy to exclude colorectal malignancy or polyps.
Enterococcal endocarditis is part of the gastrointestinal and genitourinary flora and is often implicated in patients in patients undergoing genitourinary or gastrointestinal procedures. Enterococcal endocarditis is generally difficult to treat due to high rates of antibiotic resistance and often require multi-drug regimen.
Gram negative bacilli IE is rather uncommon, the HACEK organisms (Haemophillus spp, Actinobacillus, Cardiobacterium hominis, Eikenella corrodens, Kingella spp) are responsible for approximately 3% of endocarditis cases and are the most common cause for gram negative endocarditis in non-intravenous drug users[11]. Non-HACEK organisms are a rare cause for endocarditis and only account for <1-2% of causes.
Fungal endocarditis occurs in patients who receive prolonged parenteral nutrition or antibiotics through intravenous catheters. It has also been described in intravenous drug users. Patients are often immunocompromised. The most common organisms implicated are Candida species, Histoplasma capsulatum, and Aspergillus. Mortality rates associated with fungal endocarditis exceed 80%[12].
Diagnosis
Several diagnostic criteria have been proposed for the diagnosis of IE. In clinical practice, it is the global clinical picture that leads to decision making in the diagnosis and treatment of endocarditis. The modified DUKE criteria for diagnosis is often widely used, with a sensitivity and specificity approaching ~80%.[13] The DUKE criteria divides into Definite IE, Possible IE, or Rejected IE. It uses Major criteria (microbiology, valvular abnormalities) and Minor criteria (systemic symptoms described below). Using the diagnostic criteria for IE should not override clinical judgment.
Definite IE:
- Pathologic Criteria:
- Microorganisms demonstrated by culture or histologic examination of vegetation, a vegetation that has embolized, or an intracardiac abscess specimen OR pathologic lesions; vegetation or intracardiac abscess confirmed by histologic examination showing active endocarditis
- Clinical Criteria
- 2 major criteria OR 1 major and 3 minor criteria OR 5 minor criteria
Possible IE:
- 1 Major criterion + 1 Minor criterion OR 3 minor criteria
Rejected IE:
- Firm alternate diagnosis explaining evidence of IE OR
- Resolution of IE syndrome with =<4 days of antibiotics therapy OR
- No pathologic evidence of IE at surgery or autopsy with antibiotic therapy for =<4 days OR
- Does not meet criteria for IE, as above
| Major Criteria (microbiology) | |
|---|---|
| Typical organisms x 2 blood cultures | e.g S. viridans, S. bovis, HACEK (Haemophilus spp, Aggregatibacter, Cardiobacterium hominis, Eikenella spp, Kingella kingae), S. aureus, Enterecoccus with no primary source |
| Persistent bacteremia | With blood cultures drawn >12 hours apart OR 3 out of 3, or 3 out of 4 positive blood cultures |
| Single positive blood culture for Coxiella burnetti or antiphase I IgG antibody titer >1:800 | |
| Major Criteria (valve) | |
| Echocardiogram with evidence of vegetation | TTE or TEE |
| New valvular regurgitation | |
| Minor Criteria | |
| Predisposing cardiac condition or IV drug use | |
| Fever | >38 degrees celsius or
>100.4 fahrenheit) |
| Vascular phenomena | major arterial emboli, septic pulmonary infarcts, mycotic aneurysm, intracranial hemorrhage, conjunctival hemorrhage, janeway lesions |
| Immune phenomena | glomerulonephritis, Osler nodes, Roth spots, positive RF |
| Positive blood culture not meeting above major criteria or serological evidence of active infection with organism consistent with IE | |
The sensitivity of detecting on echocardiogram varies. Transthoracic and transesophageal echocardiogram sensitivities for detecting vegetations are 50% and 90% respectively.[14][15]
| Endocarditis of prosthetic mitral valve | |
|---|---|
| PLAX: vegetations on PMVL Video courtesy: AMC Echolab, AMC, The Netherlands |
A4CH Video courtesy: AMC Echolab, AMC, The Netherlands |
| Endocarditis of the aortic valve | |
| PLAX: showing an aortic valve vegetation Video courtesy: J. Vleugels, AMC, The Netherlands |
PLAX: aortic valve vegetation Video courtesy: J. Vleugels, AMC, The Netherlands |
Therapy
Empiric therapy for infective endocarditis should only be used when there is a high index of suspicion in the absence of positive blood cultures. Three blood cultures should be drawn 30 minutes apart prior to initiating treatment. When there a high clinical probability of infective endocarditis in acute settings, empiric therapy is geared towards MRSA and coagulase negative staphylococcus. In healthcare settings and in injection drug users coverage should also include gram negative bacilli. Options for such therapy include Vancomycin + Gentamicin or, Nafcillin/Oxacillin + Tobramycin/Gentamicin.
When considering coverage for subacute endocarditis, coveraged is geared more towards streprococci spp. Options commonly include Ampicillin/Sulbactam + Gentamicin/Tobramycin, or Ceftriaxone + Vancomycin.
As soon as blood cultures become available antibiotics should be adjusted to target the identified microorganisms.
In cases of prosthetic valve endocarditis (PVE), microbiological activity depends on early (<2 months post op) or late (>2 months post op). In early PVE S.aureus accounts for 40% of the cases, followed by coagulase negative staphylococcus (17%). In late PVE coagulase negative staphylococcus accounts for 20% of cases, followed by S. aureus (18%). Coverage for enterococci, streptococci, and gram negative should be considered in empiric therapy in both groups. Rifampin + Vancomycin + Gentamicin should be initiated for PVE <12 post op. Suspected PVE >12 months post op may be treated with the same regimen as for native valves.[16]
The American Heart Association recommendation for specific antimicrobial therapy can be found in their guideline.
The European Society of Cardiology guideline for the treatment of Infective endocarditis.
Prophylaxis
According the American Heart Association guidelines published in 2007 the following groups of patients are considered to be high-risk and require prophylaxis:[17]
- Any prosthetic heart valve, or prosthetic material used for valve repair
- Previous infective endocarditis
- Congenital heart disease (CHD)
- Unrepaired cyanotic CHD, including all palliative shunts and conduits
- Completely repaired congenital heart defect with prosthetic material or device, whether placed by surgery or by catheter intervention, during the first 6 months after the procedure
- Repaired CHD with residual defects at the site or adjacent to the site of a prosthetic patch or prosthetic device (which inhibit endothelialization)
- Cardiac transplantation recipients who develop cardiac valvulopathy
- Dental procedures require prophylaxis:
- Manipulation of gingival tissue, or the periapical region of teeth or perforation of oral mucosa
- Respiratory tract procedures require prophylaxis:
- Incision or biopsy of the respiratory mucosa, or procedures involving treatment of abscess of empyema
Antibiotic prophylaxis is also recommended for any procedures on infected skin/skin structures or musculoskeletal tissue in high risk patients.[17]
Complications of Endocarditis
Common complications arising from IE can be divided into local and systemic. Local complications often arise from direct extension of the infection into cardiac structures. Systemic complications arise from embolization and bacteremias.
Heart failure occurs in 26-30% of patients with endocarditis.[18][19] It may occur acutely or over time, it is often times due to anatomical disruption from valve vegetations or destruction of nearby tissue. Development of heart failure in the setting of IE is correlated with worse outcomes. Heart failure occurs most commonly with aortic (29%) and mitral valve (20%) infections and less with tricuspid valve (8%).[12] The overall in hospital mortality rate for patients diagnosed with heart failure approaches 30%.[20]
Conduction abnormalities, commonly characterized by heart blocks in endocarditis are associated with infection extension, increased risk of embolization and increased mortality. They are reported to be present in 26%-28% of patients.[21]
Embolization is a dreaded complication of IE and most commonly affects the spleen, brain, kidneys in cases of left sided endocarditis, and the lung in right sided endocarditis. Studies report a rate of 8.5-25% and are associated with significant mortality risk.[22][23] Vegetation length, especially >10mm, infection with S. aureus, S. bovis are predictive factors for a higher rate of embolization and increased in mortality.[24] Embolization to the brain can result in mycotic aneurysms which can present with a variety of neurologic manifestations depending on the anatomic location and spread of infection in the surrounding area. Up to 30% of patients with evidence of embolization to the brain are reported to be asymptomatic.[25]
Prognosis
Prognosis of IE is largely dependent on the patient’s comorbid conditions such as diabetes,[26] hemodialysis, congestive heart failure,[27] complications of endocarditis, prosthetic valve and the microorganism identified. Generally the outcome largely depends on the organism involved. According to recent data it, the over 30 day mortality is ~15% and the 1-year mortality is ~34%.[28] Prosthetic valve endocarditis has a significant in hospital mortality of ~24%,[29] while native valve endocarditis carries a lower in hospital mortality of 12% if treated early and surgically.[30]
References
-
Cabell CH, Fowler VG, Jr, Engemann JJ, et al. "Endocarditis in the elderly: Incidence, surgery, and survival in 16,921 patients over 12 years." Circulation.2002;106(19):547.
- Miró JM, del Río A, and Mestres CA. Infective endocarditis in intravenous drug abusers and HIV-1 infected patients. Infect Dis Clin North Am. 2002 Jun;16(2):273-95, vii-viii. DOI:10.1016/s0891-5520(01)00008-3 |
- Ipek EG, Guray U, Demirkan B, Guray Y, and Aksu T. Infections of implantable cardiac rhythm devices: predisposing factors and outcome. Acta Cardiol. 2012 Jun;67(3):303-10. DOI:10.2143/AC.67.3.2160719 |
- Pappelbaum KI, Gorzelanny C, Grässle S, Suckau J, Laschke MW, Bischoff M, Bauer C, Schorpp-Kistner M, Weidenmaier C, Schneppenheim R, Obser T, Sinha B, and Schneider SW. Ultralarge von Willebrand factor fibers mediate luminal Staphylococcus aureus adhesion to an intact endothelial cell layer under shear stress. Circulation. 2013 Jul 2;128(1):50-9. DOI:10.1161/CIRCULATIONAHA.113.002008 |
- Rasmussen RV, Høst U, Arpi M, Hassager C, Johansen HK, Korup E, Schønheyder HC, Berning J, Gill S, Rosenvinge FS, Fowler VG Jr, Møller JE, Skov RL, Larsen CT, Hansen TF, Mard S, Smit J, Andersen PS, and Bruun NE. Prevalence of infective endocarditis in patients with Staphylococcus aureus bacteraemia: the value of screening with echocardiography. Eur J Echocardiogr. 2011 Jun;12(6):414-20. DOI:10.1093/ejechocard/jer023 |
- Duval X, Delahaye F, Alla F, Tattevin P, Obadia JF, Le Moing V, Doco-Lecompte T, Celard M, Poyart C, Strady C, Chirouze C, Bes M, Cambau E, Iung B, Selton-Suty C, Hoen B, and AEPEI Study Group. Temporal trends in infective endocarditis in the context of prophylaxis guideline modifications: three successive population-based surveys. J Am Coll Cardiol. 2012 May 29;59(22):1968-76. DOI:10.1016/j.jacc.2012.02.029 |
- Miro JM, Anguera I, Cabell CH, Chen AY, Stafford JA, Corey GR, Olaison L, Eykyn S, Hoen B, Abrutyn E, Raoult D, Bayer A, Fowler VG Jr, and International Collaboration on Endocarditis Merged Database Study Group. Staphylococcus aureus native valve infective endocarditis: report of 566 episodes from the International Collaboration on Endocarditis Merged Database. Clin Infect Dis. 2005 Aug 15;41(4):507-14. DOI:10.1086/431979 |
- Lalani T, Kanafani ZA, Chu VH, Moore L, Corey GR, Pappas P, Woods CW, Cabell CH, Hoen B, Selton-Suty C, Doco-Lecompte T, Chirouze C, Raoult D, Miro JM, Mestres CA, Olaison L, Eykyn S, Abrutyn E, Fowler VG Jr, and International Collaboration on Endocarditis Merged Database Study Group. Prosthetic valve endocarditis due to coagulase-negative staphylococci: findings from the International Collaboration on Endocarditis Merged Database. Eur J Clin Microbiol Infect Dis. 2006 Jun;25(6):365-8. DOI:10.1007/s10096-006-0141-z |
- McDonald JR. Acute infective endocarditis. Infect Dis Clin North Am. 2009 Sep;23(3):643-64. DOI:10.1016/j.idc.2009.04.013 |
- Das M, Badley AD, Cockerill FR, Steckelberg JM, and Wilson WR. Infective endocarditis caused by HACEK microorganisms. Annu Rev Med. 1997;48:25-33. DOI:10.1146/annurev.med.48.1.25 |
- Baddour LM, Wilson WR, Bayer AS, Fowler VG Jr, Bolger AF, Levison ME, Ferrieri P, Gerber MA, Tani LY, Gewitz MH, Tong DC, Steckelberg JM, Baltimore RS, Shulman ST, Burns JC, Falace DA, Newburger JW, Pallasch TJ, Takahashi M, Taubert KA, Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, Councils on Clinical Cardiology, Stroke, and Cardiovascular Surgery and Anesthesia, American Heart Association, and Infectious Diseases Society of America. Infective endocarditis: diagnosis, antimicrobial therapy, and management of complications: a statement for healthcare professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, and the Councils on Clinical Cardiology, Stroke, and Cardiovascular Surgery and Anesthesia, American Heart Association: endorsed by the Infectious Diseases Society of America. Circulation. 2005 Jun 14;111(23):e394-434. DOI:10.1161/CIRCULATIONAHA.105.165564 |
- Li JS, Sexton DJ, Mick N, Nettles R, Fowler VG Jr, Ryan T, Bashore T, and Corey GR. Proposed modifications to the Duke criteria for the diagnosis of infective endocarditis. Clin Infect Dis. 2000 Apr;30(4):633-8. DOI:10.1086/313753 |
- Evangelista A and Gonzalez-Alujas MT. Echocardiography in infective endocarditis. Heart. 2004 Jun;90(6):614-7. DOI:10.1136/hrt.2003.029868 |
- Mügge A, Daniel WG, Frank G, and Lichtlen PR. Echocardiography in infective endocarditis: reassessment of prognostic implications of vegetation size determined by the transthoracic and the transesophageal approach. J Am Coll Cardiol. 1989 Sep;14(3):631-8. DOI:10.1016/0735-1097(89)90104-6 |
- Habib G, Hoen B, Tornos P, Thuny F, Prendergast B, Vilacosta I, Moreillon P, de Jesus Antunes M, Thilen U, Lekakis J, Lengyel M, Müller L, Naber CK, Nihoyannopoulos P, Moritz A, Zamorano JL, and ESC Committee for Practice Guidelines. Guidelines on the prevention, diagnosis, and treatment of infective endocarditis (new version 2009): the Task Force on the Prevention, Diagnosis, and Treatment of Infective Endocarditis of the European Society of Cardiology (ESC). Endorsed by the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) and the International Society of Chemotherapy (ISC) for Infection and Cancer. Eur Heart J. 2009 Oct;30(19):2369-413. DOI:10.1093/eurheartj/ehp285 |
- Wilson W, Taubert KA, Gewitz M, Lockhart PB, Baddour LM, Levison M, Bolger A, Cabell CH, Takahashi M, Baltimore RS, Newburger JW, Strom BL, Tani LY, Gerber M, Bonow RO, Pallasch T, Shulman ST, Rowley AH, Burns JC, Ferrieri P, Gardner T, Goff D, Durack DT, American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee, American Heart Association Council on Cardiovascular Disease in the Young, American Heart Association Council on Clinical Cardiology, American Heart Association Council on Cardiovascular Surgery and Anesthesia, and Quality of Care and Outcomes Research Interdisciplinary Working Group. Prevention of infective endocarditis: guidelines from the American Heart Association: a guideline from the American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee, Council on Cardiovascular Disease in the Young, and the Council on Clinical Cardiology, Council on Cardiovascular Surgery and Anesthesia, and the Quality of Care and Outcomes Research Interdisciplinary Working Group. Circulation. 2007 Oct 9;116(15):1736-54. DOI:10.1161/CIRCULATIONAHA.106.183095 |
- Hollanders G, De Scheerder I, De Buyzere M, Ingels G, Bogaert S, and Clement DL. A six years review on 53 cases of infective endocarditis: clinical, microbiological and therapeutical features. Acta Cardiol. 1988;43(2):121-32.
- Kiefer T, Park L, Tribouilloy C, Cortes C, Casillo R, Chu V, Delahaye F, Durante-Mangoni E, Edathodu J, Falces C, Logar M, Miró JM, Naber C, Tripodi MF, Murdoch DR, Moreillon P, Utili R, and Wang A. Association between valvular surgery and mortality among patients with infective endocarditis complicated by heart failure. JAMA. 2011 Nov 23;306(20):2239-47. DOI:10.1001/jama.2011.1701 |
- Meine TJ, Nettles RE, Anderson DJ, Cabell CH, Corey GR, Sexton DJ, and Wang A. Cardiac conduction abnormalities in endocarditis defined by the Duke criteria. Am Heart J. 2001 Aug;142(2):280-5. DOI:10.1067/mhj.2001.116964 |
- Homma S and Grahame-Clarke C. Toward reducing embolic complications from endocarditis. J Am Coll Cardiol. 2003 Sep 3;42(5):781-3. DOI:10.1016/s0735-1097(03)00843-x |
- Hubert S, Thuny F, Resseguier N, Giorgi R, Tribouilloy C, Le Dolley Y, Casalta JP, Riberi A, Chevalier F, Rusinaru D, Malaquin D, Remadi JP, Ammar AB, Avierinos JF, Collart F, Raoult D, and Habib G. Prediction of symptomatic embolism in infective endocarditis: construction and validation of a risk calculator in a multicenter cohort. J Am Coll Cardiol. 2013 Oct 8;62(15):1384-92. DOI:10.1016/j.jacc.2013.07.029 |
- Thuny F, Di Salvo G, Belliard O, Avierinos JF, Pergola V, Rosenberg V, Casalta JP, Gouvernet J, Derumeaux G, Iarussi D, Ambrosi P, Calabró R, Riberi A, Collart F, Metras D, Lepidi H, Raoult D, Harle JR, Weiller PJ, Cohen A, and Habib G. Risk of embolism and death in infective endocarditis: prognostic value of echocardiography: a prospective multicenter study. Circulation. 2005 Jul 5;112(1):69-75. DOI:10.1161/CIRCULATIONAHA.104.493155 |
- Snygg-Martin U, Gustafsson L, Rosengren L, Alsiö A, Ackerholm P, Andersson R, and Olaison L. Cerebrovascular complications in patients with left-sided infective endocarditis are common: a prospective study using magnetic resonance imaging and neurochemical brain damage markers. Clin Infect Dis. 2008 Jul 1;47(1):23-30. DOI:10.1086/588663 |
- Kourany WM, Miro JM, Moreno A, Corey GR, Pappas PA, Abrutyn E, Hoen B, Habib G, Fowler VG Jr, Sexton DJ, Olaison L, Cabell CH, and ICE MD Investigators. Influence of diabetes mellitus on the clinical manifestations and prognosis of infective endocarditis: a report from the International Collaboration on Endocarditis-Merged Database. Scand J Infect Dis. 2006;38(8):613-9. DOI:10.1080/00365540600617017 |
- Nakagawa T, Wada H, Sakakura K, Yamada Y, Ishida K, Ibe T, Ikeda N, Sugawara Y, Ako J, and Momomura S. Clinical features of infective endocarditis: comparison between the 1990s and 2000s. J Cardiol. 2014 Feb;63(2):145-8. DOI:10.1016/j.jjcc.2013.06.007 |
- Bikdeli B, Wang Y, Kim N, Desai MM, Quagliarello V, and Krumholz HM. Trends in hospitalization rates and outcomes of endocarditis among Medicare beneficiaries. J Am Coll Cardiol. 2013 Dec 10;62(23):2217-26. DOI:10.1016/j.jacc.2013.07.071 |
- Wang A, Pappas P, Anstrom KJ, Abrutyn E, Fowler VG Jr, Hoen B, Miro JM, Corey GR, Olaison L, Stafford JA, Mestres CA, Cabell CH, and International Collaboration on Endocarditis Investigators. The use and effect of surgical therapy for prosthetic valve infective endocarditis: a propensity analysis of a multicenter, international cohort. Am Heart J. 2005 Nov;150(5):1086-91. DOI:10.1016/j.ahj.2005.01.023 |
- Lalani T, Cabell CH, Benjamin DK, Lasca O, Naber C, Fowler VG Jr, Corey GR, Chu VH, Fenely M, Pachirat O, Tan RS, Watkin R, Ionac A, Moreno A, Mestres CA, Casabé J, Chipigina N, Eisen DP, Spelman D, Delahaye F, Peterson G, Olaison L, Wang A, and International Collaboration on Endocarditis-Prospective Cohort Study (ICE-PCS) Investigators. Analysis of the impact of early surgery on in-hospital mortality of native valve endocarditis: use of propensity score and instrumental variable methods to adjust for treatment-selection bias. Circulation. 2010 Mar 2;121(8):1005-13. DOI:10.1161/CIRCULATIONAHA.109.864488 |