Atherosclerosis: Difference between revisions
| (21 intermediate revisions by 3 users not shown) | |||
| Line 1: | Line 1: | ||
''Ronak Delewi, MD; Hayang Yang, MsC; John Kastelein, MD, PhD''<br /><br /> | ''Ronak Delewi, MD; Hayang Yang, MsC; John Kastelein, MD, PhD''<br /><br /> | ||
{{case| | {{case| | ||
A 53 years old man, without medical history medication visits the family physician and makes an anxious impression. His friend has recently suffered from a myocardial infarction (MI) and he is worried that he might also soon face the same situation. As for family medical history, he has a father with hypertension and an uncle with diabetes mellitus. He does not seem to have any symptoms or complaints at this moment, but he has been smoking for 25 years and is overweight. Because of these characteristics he is worried that he will suffer a MI. Upon physical examination, his BMI was 29 kg/m<sup>2</sup>, RR was 152/90 mmHg and heart rate was 75 bpm. The family physician orders a blood test for lipid profile and glucose. Both turn out to be in the normal range. <br /> | A 53 years old man, without medical history or medication visits the family physician and makes an anxious impression. His friend has recently suffered from a myocardial infarction (MI) and he is worried that he might also soon face the same situation. As for family medical history, he has a father with hypertension and an uncle with diabetes mellitus. He does not seem to have any symptoms or complaints at this moment, but he has been smoking for 25 years and is overweight. Because of these characteristics he is worried that he will suffer from a MI. Upon physical examination, his BMI was 29 kg/m<sup>2</sup>, RR was 152/90 mmHg and heart rate was 75 bpm. The family physician orders a blood test for lipid profile and glucose. Both turn out to be in the normal range. <br /> | ||
The family physician gives the patient advice concerning primary prevention for atherosclerosis; quit smoking, try to achieve weight reduction, do regular physical activity, restrict alcohol consumption to less than 3 drinks a day and follow a varied and balanced diet. Regarding hypertension, the advice is to keep his RR under 140/90 mmHg. Antihypertensive medication is not indicated at this moment, because his 10-years risk of death due to cardiovascular disease (Systematic Coronary Risk Evaluation) is lower than 20%. He is advised to undergo regular checkups of cardiovascular risk profile or report to the doctor’s office in case of chest pain. | The family physician gives the patient advice concerning primary prevention for atherosclerosis; quit smoking, try to achieve weight reduction, do regular physical activity, restrict alcohol consumption to less than 3 drinks a day and follow a varied and balanced diet. Regarding hypertension, the advice is to keep his RR under 140/90 mmHg. Antihypertensive medication is not indicated at this moment, because his 10-years risk of death due to cardiovascular disease (Systematic Coronary Risk Evaluation) is lower than 20%. He is advised to undergo regular checkups of cardiovascular risk profile or report to the doctor’s office in case of chest pain. | ||
| Line 65: | Line 63: | ||
== 1.2 Arterial vessel with atherosclerosis == | == 1.2 Arterial vessel with atherosclerosis == | ||
[[File:RCA_atherosclerosis.jpg|thumb|Atheroclerotic plaque in a coronary artery]] | |||
=== Three pathologic stages of atherogenesis === | === Three pathologic stages of atherogenesis === | ||
Atherogenesis can be divided into five key steps, which are 1) endothelial dysfunction, 2) formation of lipid layer or fatty streak within the intima, 3) migration of leukocytes and smooth muscle cells | Atherogenesis can be divided into five key steps, which are 1) endothelial dysfunction, 2) formation of lipid layer or fatty streak within the intima, 3) migration of leukocytes and smooth muscle cells into the vessel wall, 4) foam cell formation and 5) degradation of extracellular matrix. Via these consecutive steps, an atherosclerotic plaque is formed. The formation of the plaque can also be divided into three major stages namely 1) the fatty streak, which represents the initiation 2) plaque progression, which represents adaption and 3) plaque disruption, which represents the clinical complication of atherosclerosis.<br /> | ||
[[File:Figure_7_-_Fatty_streak_formation_revealing_platelet_aggregation_on_the_endothelial_surface.png|right|thumb|Figure 5. Fatty streak formation]] | [[File:Figure_7_-_Fatty_streak_formation_revealing_platelet_aggregation_on_the_endothelial_surface.png|right|thumb|Figure 5. Fatty streak formation]] | ||
| Line 94: | Line 93: | ||
Endothelial dysfunction is a primary event in atherogenesis, which can be caused by various agents, such as physical stress and chemical irritants. Endothelial dysfunction is also observed in other pathological conditions, which are often related to atherosclerosis such as hypercholesterolemia, diabetes, hypertension, heart failure, cigarette smoking and aging.<br /> | Endothelial dysfunction is a primary event in atherogenesis, which can be caused by various agents, such as physical stress and chemical irritants. Endothelial dysfunction is also observed in other pathological conditions, which are often related to atherosclerosis such as hypercholesterolemia, diabetes, hypertension, heart failure, cigarette smoking and aging.<br /> | ||
Endothelial cells can display different reactions according to various levels of physical stress. There are two atheroprotective endothelial functions from physical stress. When endothelial cells are exposed to laminar flow, which | Endothelial cells can display different reactions according to various levels of physical stress. There are two atheroprotective endothelial functions from physical stress. When endothelial cells are exposed to laminar flow, which display minimal physical stress, they secrete NO. NO functions as an anti-atherosclerotic substance through vasodilation, inhibition of platelet aggregation and anti-inflammatory effects. The second function is executed, when exposed to laminar flow by an expression of the antioxidant enzyme superoxide dismutase. This enzyme performs anti-atherosclerotic role by acting against reactive oxygen species, which are produced by chemical irritants or transient ischemia in the vessel.<br /> | ||
{| class="wikitable" border="1" style='float: right' | {| class="wikitable" border="1" style='float: right' | ||
|- | |- | ||
| Line 108: | Line 107: | ||
|}<br /> | |}<br /> | ||
Unfortunately, these two atheroprotective endothelial functions can be impaired by several factors. The first factor is disturbed flow (low shear stress with rapid | Unfortunately, these two atheroprotective endothelial functions can be impaired by several factors. The first factor is disturbed flow (low shear stress with rapid fluctuations), which is typically located at arterial branch points and bifurcations and can impair the protective functions. This is well illustrated by the difference in prevalence of atherosclerosis between branched arteries and bifurcated vessels. Bifurcation areas such as the common carotid and left coronary arteries are common deposition sites for atherosclerosis than arteries with few branches such as the internal mammary artery. Thus, many observations show that the distribution of atherosclerotic lesions is common in large vessels and they vary in location and frequency among different vascular beds. These findings encourage a belief that hemodynamic factors play an important role in atherogenesis. Furthermore, the fact that hypertension intensifies the severity of atherosclerotic lesions additionally supports this hypothesis.<br /> | ||
[[File:Figure_8_-_Endothelial_dysfunction_-_Leukocyte_adhesion_and_migration_into_the_deep_layer_of_the_intima.png|thumb|left|Figure 8. Endothelial dysfunction: Leukocyte adhesion and migration into the deep layer of the intima.]]<br /> | [[File:Figure_8_-_Endothelial_dysfunction_-_Leukocyte_adhesion_and_migration_into_the_deep_layer_of_the_intima.png|thumb|left|Figure 8. Endothelial dysfunction: Leukocyte adhesion and migration into the deep layer of the intima.]]<br /> | ||
| Line 135: | Line 134: | ||
==== ''Smooth muscle cell migration'' ==== | ==== ''Smooth muscle cell migration'' ==== | ||
Smooth muscle cells play a central role at the phase of transition from fatty streak to plaque formation. During this phase, smooth muscle cells migrate from the media to the intima. After migration, smooth muscle cells proliferate within the intima and secrete extracellular matrix macromolecules. Additionally, foam cells, activated platelets and endothelium stimulate substances that induce the migration and accumulation of smooth muscle cells. For example, foam cells release platelet derived growth factor (PDGF), cytokines and growth factors that directly contribute to the migration and proliferation process, and they also activate smooth muscle cells and leukocytes to reinforce inflammation in the atherosclerotic lesion. Although plaque progression is traditionally known as a gradual and continuous process, recent evidence claims that this process can be strongly accentuated by bursts of smooth muscle replication. The observation of small ruptures within the plaque occurring without any clinical symptoms or signs supports this suggestion. These small ruptures expose tissue factor secreted by foam cells that stimulates coagulation and microthrombus formation in the lesion. Such | Smooth muscle cells play a central role at the phase of transition from fatty streak to plaque formation. During this phase, smooth muscle cells migrate from the media to the intima. After migration, smooth muscle cells proliferate within the intima and secrete extracellular matrix macromolecules. Additionally, foam cells, activated platelets and endothelium stimulate substances that induce the migration and accumulation of smooth muscle cells. For example, foam cells release platelet derived growth factor (PDGF), cytokines and growth factors that directly contribute to the migration and proliferation process, and they also activate smooth muscle cells and leukocytes to reinforce inflammation in the atherosclerotic lesion. Although plaque progression is traditionally known as a gradual and continuous process, recent evidence claims that this process can be strongly accentuated by bursts of smooth muscle replication. The observation of small ruptures within the plaque occurring without any clinical symptoms or signs supports this suggestion. These small ruptures expose tissue factor secreted by foam cells that stimulates coagulation and microthrombus formation in the lesion. Such microthrombi contain activated platelets that release additional factors such as PDGF and heparinase that can further stimulate local smooth muscle cell migration and proliferation. Heparinase stimulates smooth muscle cell migration and proliferation by degrading heparan sulfate, which normally counteracts this process.<br /> | ||
==== ''Extracellular matrix metabolism'' ==== | ==== ''Extracellular matrix metabolism'' ==== | ||
Metabolic processes in extracellular matrix | Metabolic processes in extracellular matrix play a central role in bridging the plaque progression to plaque rupture. Ultimately, this process weakens the fibrous cap, predisposing it to rupture. This process is influenced by the balance of matrix deposition synthesis by smooth muscle cells and degradation by matrix metalloproteinases (MMP), a class of proteolytic enzymes. For example, PDGF and TGF-β stimulate interstitial collagen production, while inflammatory cytokines such as IFN-γ inhibits collagen synthesis. TGF-β also induces formation of fibronectin and proteoglycans. It is an important regulator since it enhances the expression of protease inhibitors, leading to the inhibition of proteolytic enzymes that promote matrix degradation. On the other hand, inflammatory cytokines weaken the fibrous cap by stimulating local foam cells to secrete MMP that degrades collagen and elastin of the fibrous cap. Furthermore, the deeper parts of the thickened intima undergo necrosis due to poor nourishment.<br /> | ||
=== Plaque rupture === | === Plaque rupture === | ||
| Line 153: | Line 152: | ||
The concept of ‘vulnerable plaque’ has developed into a new concept of ‘vulnerable patient’ as the concept of pathogenesis of atherosclerosis was linked to a person’s susceptibility to coagulation and thus vascular events, which can be influenced by many personal factors such as genetics (e.g. procoagulant prothombin gene mutation), coexisting condition (e.g. diabetes), and lifestyle factors (e.g. smoking, obesity).<br /> | The concept of ‘vulnerable plaque’ has developed into a new concept of ‘vulnerable patient’ as the concept of pathogenesis of atherosclerosis was linked to a person’s susceptibility to coagulation and thus vascular events, which can be influenced by many personal factors such as genetics (e.g. procoagulant prothombin gene mutation), coexisting condition (e.g. diabetes), and lifestyle factors (e.g. smoking, obesity).<br /> | ||
{| | |||
|- | |||
| rowspan="2" | [[File:plaque_rupture_A.svg|100px]] | |||
| rowspan="2" | [[File:plaque_rupture_B.svg|100px]] | |||
| rowspan="2" | [[File:split_arrow.svg|50px]] | |||
| [[File:plaque_rupture_C.svg|100px]] || [[File:plaque_rupture_clot.svg|100px]] | |||
|- | |||
| [[File:plaque_rupture_D.svg|100px]] | |||
|- | |||
| colspan="4" width="450px" | Progression of coronary atherosclerosis can be gradual (bottom) or can lead to plaque rupture with acute occlusion of a coronary vessel due to clot formation | |||
|} | |||
== Complications of atherosclerosis == | == Complications of atherosclerosis == | ||
| Line 170: | Line 181: | ||
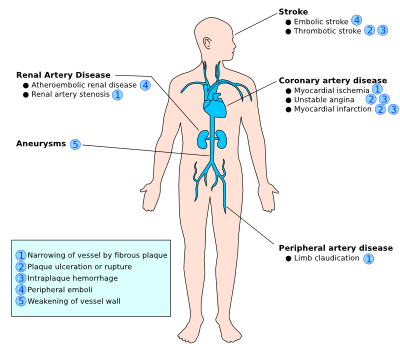
The clinical complications of atherosclerosis are highly dependent on the location and size of affected vessels, the duration of the chronic process, and the type of plaque, since the severity of impairment of atherosclerosis differs throughout the vasculature. For example, ‘stable plaque’ can easily result into angina pectoris due to its thick fibrous cap that directly affects the diameter of the relatively small coronary vessels. On the other hand, ‘vulnerable plaque’ is non-stenotic, but can easily cause acute thrombosis and therefore myocardial infarction due to its fragility towards rupture when located at physically stressed areas such as bifurcations. Often with ‘vulnerable plaques’ there are relatively few symptoms, however they are more numerous and dispersed throughout the arteries compared to ‘stable plaque’. Thus, you can either have an occlusion due to the growing plaque or due to the embolization of the ruptured fragments of the original plaque. Due to the difficult detection of ‘vulnerable plaques’ while they are widely dispersed, it is highly important to tackle the risk factors prior to plaque rupture. Thus in the following paragraph, we will highlight the clinical risk factors associated with atherosclerosis. The four major clinical consequences of atherosclerosis are listed and explained below.<br /> | The clinical complications of atherosclerosis are highly dependent on the location and size of affected vessels, the duration of the chronic process, and the type of plaque, since the severity of impairment of atherosclerosis differs throughout the vasculature. For example, ‘stable plaque’ can easily result into angina pectoris due to its thick fibrous cap that directly affects the diameter of the relatively small coronary vessels. On the other hand, ‘vulnerable plaque’ is non-stenotic, but can easily cause acute thrombosis and therefore myocardial infarction due to its fragility towards rupture when located at physically stressed areas such as bifurcations. Often with ‘vulnerable plaques’ there are relatively few symptoms, however they are more numerous and dispersed throughout the arteries compared to ‘stable plaque’. Thus, you can either have an occlusion due to the growing plaque or due to the embolization of the ruptured fragments of the original plaque. Due to the difficult detection of ‘vulnerable plaques’ while they are widely dispersed, it is highly important to tackle the risk factors prior to plaque rupture. Thus in the following paragraph, we will highlight the clinical risk factors associated with atherosclerosis. The four major clinical consequences of atherosclerosis are listed and explained below.<br /> | ||
# Acute narrowing of the vessel lumen: When the plaque ruptures, it will release its pro-coagulants in the bloodstream and that will lead to the formation of thrombus at the rupture site. The rupture often occurs at sites of erosion and fissuring on the fibrous cap surface. This thrombus may cause a complete occlusion of a particular vessel and result in ischemic necrosis (infarction) of the tissue that this particular vessel is supplying to. Clinically this is manifested as stroke, MI, gangrene of several possible organs such as intestine, spleen or lower extremities. These | # Acute narrowing of the vessel lumen: When the plaque ruptures, it will release its pro-coagulants in the bloodstream and that will lead to the formation of thrombus at the rupture site. The rupture often occurs at sites of erosion and fissuring on the fibrous cap surface. This thrombus may cause a complete occlusion of a particular vessel and result in ischemic necrosis (infarction) of the tissue that this particular vessel is supplying to. Clinically this is manifested as stroke, MI, gangrene of several possible organs such as intestine, spleen or lower extremities. These occlusions may also dissolve spontaneously due to pro-fibrinolytic enzymes such as streptokinase and tissue plasminogen activator (TPA). | ||
# Chronic occlusion: When the occlusion is gradual and incomplete, it may chronically disturb the blood supply to tissues in the distribution of the affected vessel. This can result in chronic ischemia of those tissues that can additionally lead to complaints of angina pectoris or intermittent claudication or to organ atrophy (e.g. atrophy of kidney, intestines and skin due to impairment of blood flow in renal artery, mesenteric artery, peripheral vasculature among diabetics.). | # Chronic occlusion: When the occlusion is gradual and incomplete, it may chronically disturb the blood supply to tissues in the distribution of the affected vessel. This can result in chronic ischemia of those tissues that can additionally lead to complaints of angina pectoris or intermittent claudication or to organ atrophy (e.g. atrophy of kidney, intestines and skin due to impairment of blood flow in renal artery, mesenteric artery, peripheral vasculature among diabetics.). | ||
# Embolism: Embolization is the transfer of the fragments of disrupted atheroma to distal vascular sites, which results into occlusion of those sites. For example, fragments of thrombi in abdominal aorta may transfer to the popliteal artery subsequently resulting in gangrene of the leg. Ulceration of atheroma may also produce ‘cholesterol crystal emboli’. This type of emboli is visualized as needle-shaped areas in affected tissues, mostly detected in the kidney. | # Embolism: Embolization is the transfer of the fragments of disrupted atheroma to distal vascular sites, which results into occlusion of those sites. For example, fragments of thrombi in abdominal aorta may transfer to the popliteal artery subsequently resulting in gangrene of the leg. Ulceration of atheroma may also produce ‘cholesterol crystal emboli’. This type of emboli is visualized as needle-shaped areas in affected tissues, mostly detected in the kidney. | ||
# Aneurysm: After a chronic period, atherosclerotic lesion may extend into the medial layer, resulting into atrophy and loss of elastic tissue. This can subsequently cause dilatation and weakness of the artery, forming aneurysm. Over time, | # Aneurysm: After a chronic period, atherosclerotic lesion may extend into the medial layer, resulting into atrophy and loss of elastic tissue. This can subsequently cause dilatation and weakness of the artery, forming aneurysm. Over time, aneurysms may suddenly rupture and result in a life-threatening situation for the patients. | ||
== Risk factors of atherosclerosis == | == Risk factors of atherosclerosis == | ||
| Line 228: | Line 239: | ||
In primary prevention, pharmacologic agents are the second option when lifestyle modifications fail to achieve targeted lipid profile. There are several groups of lipid-altering medicines such as HMG-CoA reductase inhibitors (statins), niacin, fibric acid derivatives, cholesterol intestinal absorption inhibitors, and bile acid-binding agents. In the clinical setting, statins are widely used, being the most cost-effective LDL-lowering drugs. They reduce intracellular cholesterol concentration by inhibiting HMG-CoA reductase, which is an enzyme that synthesizes cholesterol. This results into increased LDL-receptor expression and therefore leads to higher clearance of LDL molecules from blood. They also affect the liver and thereby lower the rate of VLDL synthesis, which results into lower levels of serum triglyceride. Statins also raise HDL, but this mechanism is not fully understood yet. | |||
In primary prevention, pharmacologic agents are the second option when lifestyle modifications fail to achieve targeted lipid profile. There are several groups of lipid-altering medicines such as HMG-CoA reductase inhibitors ('''statins'''), niacin, fibric acid derivatives, cholesterol intestinal absorption inhibitors, and bile acid-binding agents. In the clinical setting, statins are widely used, being the most cost-effective LDL-lowering drugs. They reduce intracellular cholesterol concentration by inhibiting HMG-CoA reductase, which is an enzyme that synthesizes cholesterol. This results into increased LDL-receptor expression and therefore leads to higher clearance of LDL molecules from blood. They also affect the liver and thereby lower the rate of VLDL synthesis, which results into lower levels of serum triglyceride. Statins also raise HDL, but this mechanism is not fully understood yet. | |||
| Line 236: | Line 248: | ||
Inhibiting HMG-CoA reductase results in several mechanisms that explain the beneficial effect of using statins. One beneficial mechanism is via lowering LDL and raising HDL. This results into less lipid content in atherosclerotic plaques and improve their biologic activity. Furthermore, the anti-thrombotic and anti-inflammatory profile is enhanced by other mechanisms such as increased NO synthesis and fibrinolytic activity, inhibition of smooth muscle proliferation and monocyte recruitment, and reduced production of matrix-degrading enzymes by macrophages. Several studies suggest that other mechanisms also contribute to the anti-inflammatory profile. For example, statins reduce endothelial expression of leukocyte adhesion molecules and macrophage tissue factor production by inhibiting the macrophage cytokines or by activating PPAR-α. Another anti-inflammatory action of statins, supported by clinical trials is reducing the serum level of C-reactive protein, which is a marker of inflammation. <br /> | Inhibiting HMG-CoA reductase results in several mechanisms that explain the beneficial effect of using statins. One beneficial mechanism is via lowering LDL and raising HDL. This results into less lipid content in atherosclerotic plaques and improve their biologic activity. Furthermore, the anti-thrombotic and anti-inflammatory profile is enhanced by other mechanisms such as increased NO synthesis and fibrinolytic activity, inhibition of smooth muscle proliferation and monocyte recruitment, and reduced production of matrix-degrading enzymes by macrophages. Several studies suggest that other mechanisms also contribute to the anti-inflammatory profile. For example, statins reduce endothelial expression of leukocyte adhesion molecules and macrophage tissue factor production by inhibiting the macrophage cytokines or by activating PPAR-α. Another anti-inflammatory action of statins, supported by clinical trials is reducing the serum level of C-reactive protein, which is a marker of inflammation. <br /> | ||
Despite the effectiveness of statins, niacin, fibric acid derivatives, cholesterol intestinal absorption inhibitors, and bile acid-binding agents in managing serum lipid levels, further advancements in pharmacologic lipid management have introduced additional options for reducing cardiovascular risk associated with atherosclerosis. Two notable additions to the lipid-lowering arsenal are PCSK9 inhibitors and bempedoic acid. | |||
Proprotein convertase subtilisin/kexin type 9 '''(PCSK9) inhibitors''' are a novel class of medications that significantly lower low-density lipoprotein cholesterol (LDL-C). PCSK9 is a protein that binds to LDL receptors on the liver surface, leading to their degradation and preventing them from removing LDL cholesterol from the blood. By inhibiting PCSK9, these drugs increase the number of LDL receptors available to clear LDL from the blood, thus lowering LDL-C levels. | |||
Clinical trials have demonstrated that PCSK9 inhibitors, such as evolocumab and alirocumab, can reduce LDL-C levels by up to 60% when used alone or in combination with statins. This significant reduction in LDL-C levels has been associated with a decreased risk of cardiovascular events, making PCSK9 inhibitors an important option for patients who are statin-intolerant or for whom statins alone are insufficient to achieve LDL-C targets. | |||
'''Bempedoic acid''' represents another innovative approach to lowering LDL-C levels. It acts by inhibiting ATP citrate lyase, an enzyme upstream of HMG-CoA reductase in the cholesterol biosynthesis pathway. This inhibition leads to reduced hepatic cholesterol synthesis and upregulation of LDL receptors, resulting in decreased LDL-C levels. Bempedoic acid has the advantage of being metabolized in the liver, sparing muscle tissue and potentially offering a safer alternative for patients who experience muscle-related side effects with statins. | |||
In clinical trials, bempedoic acid has been shown to lower LDL-C levels by up to 20% when used as monotherapy and has shown additional LDL-C lowering when used in combination with statins. Furthermore, bempedoic acid has been associated with reductions in biomarkers of inflammation, such as high-sensitivity C-reactive protein (hsCRP), suggesting potential anti-inflammatory benefits beyond its lipid-lowering effects. | |||
==== ''Tobacco smoking'' ==== | ==== ''Tobacco smoking'' ==== | ||
Tobacco use is known to increase the risk of atherosclerosis and ischemic heart disease based on numerous studies. For example, INTERHEART study shows that smoking is responsible for 36% of the population-attributable risk of a first MI. Other studies showed that smoking is an independent major risk factor for coronary heart disease, cerebrovascular disease and total atherosclerotic cardiovascular disease. The Atherosclerosis Risk in Communities Study measured the direct effect of smoking on the development of atherosclerosis. They measured intima-medial thickness of the carotid artery of 10,914 patients for three years with ultrasound. Their result showed that current smokers had a 50% increased progression of atherosclerosis in comparison to nonsmokers during the study period. Also patients with environmental tobacco smoke exposure (passive smokers) had 20% higher rate of atherosclerotic progress versus patients without environmental smoke exposure.<br /> | Tobacco use, including environmental smoking exposure, is known to increase the risk of atherosclerosis and ischemic heart disease based on numerous studies. For example, INTERHEART study shows that smoking is responsible for 36% of the population-attributable risk of a first MI. Other studies showed that smoking is an independent major risk factor for coronary heart disease, cerebrovascular disease and total atherosclerotic cardiovascular disease. The Atherosclerosis Risk in Communities Study measured the direct effect of smoking on the development of atherosclerosis. They measured intima-medial thickness of the carotid artery of 10,914 patients for three years with ultrasound. Their result showed that current smokers had a 50% increased progression of atherosclerosis in comparison to nonsmokers during the study period. Also patients with environmental tobacco smoke exposure (passive smokers) had 20% higher rate of atherosclerotic progress versus patients without environmental smoke exposure.<br /> | ||
Tobacco smoking can lead to many mechanisms that contribute to atherosclerosis. Smoking also leads to increased LDL levels, decreased HDL levels in blood and elevated insulin resistance. In addition it enhances oxidative modification of LDL by releasing free radicals and reduces generation of nitric oxide. This can promote endothelial dysfunction and thus lead to impairment of vasodilatation of coronary arteries and reduction of coronary flow reserve even in passive smokers. Tobacco smoking inappropriately stimulates sympathetic nervous system, increasing heart rate, blood pressure and perhaps coronary vasoconstriction. Smoking promotes a prothrombotic environment through inhibition of endothelial release of tissue plasminogen activator, elevation of fibrinogen concentration in blood, enhancement of platelet activity (possibility related to sympathetic activation) and enhanced expression of tissue factor. Smoking can even damage the vessel wall and ultimately cause a decrease in the elasticity | Tobacco smoking can lead to many mechanisms that contribute to atherosclerosis. Smoking also leads to increased LDL levels, decreased HDL levels in blood and elevated insulin resistance. In addition it enhances oxidative modification of LDL by releasing free radicals and reduces generation of nitric oxide. This can promote endothelial dysfunction and thus lead to impairment of vasodilatation of coronary arteries and reduction of coronary flow reserve even in passive smokers. Tobacco smoking inappropriately stimulates sympathetic nervous system, increasing heart rate, blood pressure and perhaps coronary vasoconstriction. Smoking promotes a prothrombotic environment through inhibition of endothelial release of tissue plasminogen activator, elevation of fibrinogen concentration in blood, enhancement of platelet activity (possibility related to sympathetic activation) and enhanced expression of tissue factor. Smoking can even damage the vessel wall and ultimately cause a decrease in the elasticity of the artery, enhancing the stiffness of vessel wall. Smoking has been associated with increased C-reactive protein and fibrinogen, suggesting a correlation with inflammatory response, which is an important part of atherogenesis. There have also been findings that show higher expression of leukocyte adhesion molecules among smokers than nonsmokers. Smoking may additionally induce tissue hypoxia through displacement of oxygen with carbon monoxide in hemoglobin. <br /> | ||
To stop smoking is known as one of the most effective preventive measures of CVD and their complications. Soon after cessation, cardiac risks due to smoking decreases in a short period, and continues to diminish when cessation is permanently preserved. The risk for cardiovascular disease among patients with coronary heart disease decreases 7-47%. Not only does cessation of smoking reduce risk of CVD, but also substantially reduce the risk of all-cause mortality.<br /> | To stop smoking is known as one of the most effective preventive measures of CVD and their complications. Soon after cessation, cardiac risks due to smoking decreases in a short period, and continues to diminish when cessation is permanently preserved. The risk for cardiovascular disease among patients with coronary heart disease decreases 7-47%. Not only does cessation of smoking reduce risk of CVD, but also substantially reduce the risk of all-cause mortality.<br /> | ||
| Line 294: | Line 312: | ||
==== ''Diet'' ==== | ==== ''Diet'' ==== | ||
A healthy diet reduces CVD risk. In general, when following the rules for a healthy diet, no dietary supplements are needed. N-3 polyunsaturated fatty acid (PUFA) consumption mainly from oily fish, is potentially associated with beneficial effects on cardiac risk factors, notably reduction in triglycerides but not all randomized, controlled trials have shown reductions in CV events Thus current recommendations are to increase PUFA intake through fish consumption, rather than from supplements. Recently, the largest study ever conducted with a so-called ‘Mediterranean’ diet, supplemented with extra-virgin olive oil or nuts, reduced the incidence of major cardiovascular events in patients at high risk of CV events but without prior CV disease.<cite>Estruch</cite> | |||
==== ''Alcohol consumption'' ==== | ==== ''Alcohol consumption'' ==== | ||
| Line 354: | Line 372: | ||
<br /> | <br /> | ||
Antihypertensive therapy can either consist of lifestyle interventions or pharmacotherapy. Lifestyle modifications consist of diet, body weight reduction, increased activity, and cessation of smoking. As for diet, high consumption of fruits, vegetables, dairy products low in fat, fish oils, potassium and reduced consumption of sodium and alcohol are recommended. | Antihypertensive therapy can either consist of lifestyle interventions or pharmacotherapy. Lifestyle modifications consist of diet, body weight reduction, increased activity, and cessation of smoking. As for diet, high consumption of fruits, vegetables, dairy products low in fat, fish oils, potassium and reduced consumption of sodium and alcohol are recommended. | ||
The indication for pharmacotherapy depends on the severity of hypertension and on the assessment of total CVD risk. Several large trials have shown that pharmacotherapy for hypertension can substantially reduce major cardiovascular events such as MI and stroke. Drug therapy is indicated when chronic SBP ≥ 160mmHg and/or DBP ≥100mmHg, or if target organ damage is present. | The indication for pharmacotherapy depends on the severity of hypertension and on the assessment of total CVD risk. Several large trials have shown that pharmacotherapy for hypertension can substantially reduce major cardiovascular events such as MI and stroke. Drug therapy is indicated when chronic SBP ≥ 160mmHg and/or DBP ≥100mmHg, or if target organ damage is present. | ||
==== ''Diabetes Mellitus'' ==== | |||
With estimated global incidence of 170 million people, diabetes mellitus is a large problem worldwide. Diabetes mellitus increases the risk of acute coronary events by three- to five folds and 80% of diabetic patients will face atherosclerosis-related cardiovascular diseases. Risk for atherosclerosis among diabetics is considered to be as high as | With estimated global incidence of 170 million people, diabetes mellitus is a large problem worldwide. Diabetes mellitus increases the risk of acute coronary events by three- to five folds and 80% of diabetic patients will face atherosclerosis-related cardiovascular diseases. Risk for atherosclerosis among diabetics is considered to be as high as in patients with previous MI. Based on this observation, the National Cholesterol Education Program report from the United States and guidelines from Europe considers type 2 diabetes to be a CHD equivalent, categorizing it to the highest risk for MI.<br /> | ||
There are several possible mechanisms that make this group particularly vulnerable to atherosclerosis. An example of mechanism is non-enzymatic glycation of lipoproteins, which promotes uptake of cholesterol by scavenger macrophages. Furthermore, pro-thrombotic and anti-fibrinolytic properties of diabetes can also contribute to this vulnerability. The high prevalence of endothelial dysfunction among diabetes group leads to reduced bioavailability of NO and enhanced leukocyte adhesion. The most effective prevention of atherosclerosis among diabetes group is tight regulation of serum glucose levels. This intervention significantly reduces the risk of microvascular complications such as retinopathy and nephropathy. Furthermore, intense anti-diabetic regime also reduced macrovascular outcomes such as MI and stroke among a group of diabetes type 1. Additionally managing hypertension and dyslipidemia among diabetic groups also significantly reduces the risk of cardiovascular diseases.<br /> | There are several possible mechanisms that make this group particularly vulnerable to atherosclerosis. An example of mechanism is non-enzymatic glycation of lipoproteins, which promotes uptake of cholesterol by scavenger macrophages. Furthermore, pro-thrombotic and anti-fibrinolytic properties of diabetes can also contribute to this vulnerability. The high prevalence of endothelial dysfunction among diabetes group leads to reduced bioavailability of NO and enhanced leukocyte adhesion. The most effective prevention of atherosclerosis among diabetes group is tight regulation of serum glucose levels. This intervention significantly reduces the risk of microvascular complications such as retinopathy and nephropathy. Furthermore, intense anti-diabetic regime also reduced macrovascular outcomes such as MI and stroke among a group of diabetes type 1. Additionally managing hypertension and dyslipidemia among diabetic groups also significantly reduces the risk of cardiovascular diseases.<br /> | ||
== References == | == References == | ||
<biblio> | |||
#Anderson Anderson J.L., Carlquist J.F., Muhlestein J.B., et al. “Evaluation of C-reactive protein, an inflammatory marker, and infectious serology as risk factors for coronary artery disease and myocardial infarction.” J Am Coll Cardiol 1998; 32:35. | |||
#Barua Barua R.S., Ambrose J.A., Eales-Reynolds L.J., et al. “Dysfunctional endothelial nitric oxide biosynthesis in healthy smokers with impaired endothelium-dependent vasodilatation.” Circulation 2001; 104:1905. | |||
#Barter Barter P.J., Caulfield M., Eriksson M., et al. “Effects of torcetrapib in patients at high risk for coronary events.” N Engl J Med 2007; 357:2109. | |||
#Bazzano Bazzano L.A., He J., Muntner P., et al. “Relationship between cigarette smoking and novel risk factors for cardiovascular disease in the United States.” Ann Intern Med 2003; 138:891. | |||
#Bennet Bennet A., Di Angelantonio E., Erqou S., et al. “Lipoprotein(a) levels and risk of future coronary heart disease: large-scale prospective data.” Arch Intern Med 2008; 168:598. | |||
#Turnbull Blood Pressure Lowering Treatment Trialists' Collaboration, Turnbull F., Neal B., et al. “Effects of different regimens to lower blood pressure on major cardiovascular events in older and younger adults: meta-analysis of randomised trials.” BMJ 2008; 336:1121. | |||
#Burke Burke A.P., Farb A., Malcom G.T., Liang Y., Smialek J.E., Virmani R. "Plaque rupture and sudden death related to exertion in men with coronary artery disease." JAMA. 1999;281(10):921. | |||
#Camm Camm A.J., Füscher T.F., Serruys P.W., eds. (2010). The ESC Textbook of Cardiovascular Medicine Online Second Edition. Oxford: Oxford University Press. | |||
#Celermajer Celermajer D.S., Sorensen K.E., Georgakopoulos D., et al. “Cigarette smoking is associated with dose-related and potentially reversible impairment of endothelium-dependent dilation in healthy young adults.” Circulation 1993; 88:2149. | |||
#Dauchet Dauchet L., Amouyel P., Dallongeville J. “Fruit and vegetable consumption and risk of stroke: a meta-analysis of cohort studies.” Neurology 2005; 65:1193. | |||
#DeBacker De Backer G., Ambrosioni E., Borch-Johnsen K., et al. “European guidelines on cardiovascular disease prevention in clinical practice: third joint task force of European and other societies on cardiovascular disease prevention in clinical practice (constituted by representatives of eight societies and by invited experts).” Eur J Cardiovasc Prev Rehabil 2003; 10:S1. | |||
#Downs Downs J.R., Clearfield M., Weis S., Whitney E., Shapiro D.R., Beere P.A., Langendorfer A., Stein EA, Kruyer W, Gotto AM Jr. "Primary prevention of acute coronary events with lovastatin in men and women with average cholesterol levels: results of AFCAPS/TexCAPS. Air Force/Texas Coronary Atherosclerosis Prevention Study." JAMA. 1998;279(20):1615. | |||
#Eckel Eckel R.H., York D.A., Rössner S., et al. “Prevention Conference VII: Obesity, a worldwide epidemic related to heart disease and stroke: executive summary.” Circulation 2004; 110:2968. | |||
#Erqou Emerging Risk Factors Collaboration, Erqou S., Kaptoge S., et al. “Lipoprotein(a) concentration and the risk of coronary heart disease, stroke, and nonvascular mortality.” JAMA 2009; 302:412. | |||
#Fuchs Fuchs C.S., Stampfer M.J., Colditz G.A., et al. “Alcohol consumption and mortality among women.” N Engl J Med 1995; 332:1245. | |||
#Grady Grady D., Herrington D., Bittner V., et al. “Cardiovascular disease outcomes during 6.8 years of hormone therapy: Heart and Estrogen/progestin Replacement Study follow-up (HERS II).” JAMA 2002; 288:49. | |||
#Harker Harker L.A., Ross R., Slichter S.J., Scott C.R. “Homocystine-induced arteriosclerosis. The role of endothelial cell injury and platelet response in its genesis.” J Clin Invest 1976; 58:731. | |||
#Haffner Haffner S.M., Lehto S., Rönnemaa T., et al. “Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with and without prior myocardial infarction.” N Engl J Med 1998; 339:229. | |||
#Howard Howard G., Wagenknecht L.E., Burke G.L., Diez-Roux A., Evans G.W., McGovern P., Nieto F.J., Tell G.S. "Cigarette smoking and progression of atherosclerosis: The Atherosclerosis Risk in Communities (ARIC) Study." JAMA. 1998;279(2):119. | |||
#Jackson Jackson R., Scragg R., Beaglehole R. “Alcohol consumption and risk of coronary heart disease.” BMJ 1991; 303:211. | |||
#Jee Jee S.H., Suh I., Kim I.S., Appel L.J. "Smoking and atherosclerotic cardiovascular disease in men with low levels of serum cholesterol: the Korea Medical Insurance Corporation Study." JAMA. 1999;282(22):2149. | |||
#Kannel Kannel W.B., D'Agostino R.B., Belanger A.J. “Fibrinogen, cigarette smoking, and risk of cardiovascular disease: insights from the Framingham Study.” Am Heart J 1987; 113:1006. | |||
#Klein Klein S., Burke L.E., Bray G.A., et al. “Clinical implications of obesity with specific focus on cardiovascular disease: a statement for professionals from the American Heart Association Council on Nutrition, Physical Activity, and Metabolism: endorsed by the American College of Cardiology Foundation.” Circulation 2004; 110:2952. | |||
#Knoops Knoops K.T., de Groot L.C., Kromhout D., et al. “Mediterranean diet, lifestyle factors, and 10-year mortality in elderly European men and women: the HALE project.” JAMA 2004; 292:1433. | |||
#VanZuiden Kwaliteitsinstituut voor de gezondheidszorg CBO and Nederlandse Huisartsen Genootschap. (2006). Multidisciplinaire richtlijn cardiovasculair risicomanagement 2006. Alphen aan den Rijn: Van Zuiden Communications. | |||
#Lilly Lilly L.S. (ed.). (2007). Pathophysiology of Heart Disease. Baltimore: Lippincott Williams & Wilkins, p. 118–140. | |||
#NCEP Lilly L.S. (ed.). (2007). Pathophysiology of Heart Disease. Baltimore: Lippincott Williams & Wilkins, p. 118–140. | |||
#Nordmann Nordmann A.J., Suter-Zimmermann K., Bucher H.C., et al. “Meta-analysis comparing Mediterranean to low-fat diets for modification of cardiovascular risk factors.” Am J Med 2011; 124:841. | |||
#Mancia Mancia G., Messerli F., Bakris G., et al. “Blood pressure control and improved cardiovascular outcomes in the International Verapamil SR-Trandolapril Study.” Hypertension 2007; 50:299. | |||
#McCully McCully K.S. “Vascular pathology of homocysteinemia: implications for the pathogenesis of arteriosclerosis.” Am J Pathol 1969; 56:111. | |||
#Mendis Mendis S., Puska P., Norrving B. (eds.). (2011). Global Atlas on Cardiovascular Disease Prevention and Control. Geneva: World Health Organization. | |||
#Mukamal Mukamal K.J., Chung H., Jenny N.S., et al. “Alcohol consumption and risk of coronary heart disease in older adults: the Cardiovascular Health Study.” J Am Geriatr Soc 2006; 54:30. | |||
#Newby Newby D.E., Wright R.A., Labinjoh C., et al. “Endothelial dysfunction, impaired endogenous fibrinolysis, and cigarette smoking: a mechanism for arterial thrombosis and myocardial infarction.” Circulation 1999; 99:1411. | |||
#O'Keefe O'Keefe J.H., Bybee K.A., Lavie C.J. “Alcohol and cardiovascular health: the razor-sharp double-edged sword.” J Am Coll Cardiol 2007; 50:1009. | |||
#Paffenbarger Paffenbarger R.S. Jr, Hyde R.T., Wing A.L., et al. “The association of changes in physical-activity level and other lifestyle characteristics with mortality among men.” N Engl J Med 1993; 328:538. | |||
#Pasceri Pasceri V., Cheng J.S., Willerson J.T., et al. “Modulation of C-reactive protein-mediated monocyte chemoattractant protein-1 induction in human endothelial cells by anti-atherosclerosis drugs.” Circulation 2001; 103:2531. | |||
#Pasceri2 Pasceri V., Willerson J.T., Yeh E.T. “Direct proinflammatory effect of C-reactive protein on human endothelial cells.” Circulation 2000; 102:2165. | |||
#Pearson Pearson T.A., Mensah G.A., Alexander R.W., et al. “Markers of inflammation and cardiovascular disease: application to clinical and public health practice: A statement for healthcare professionals from the Centers for Disease Control and Prevention and the American Heart Association.” Circulation 2003; 107:499. | |||
#Poirier Poirier P., Giles T.D., Bray G.A., et al. “Obesity and cardiovascular disease: pathophysiology, evaluation, and effect of weight loss: an update of the 1997 American Heart Association Scientific Statement on Obesity and Heart Disease from the Obesity Committee of the Council on Nutrition, Physical Activity, and Metabolism.” Circulation 2006; 113:898. | |||
#Powell Powell K.E., Thompson P.D., Caspersen C.J., Kendrick J.S. “Physical activity and the incidence of coronary heart disease.” Annu Rev Public Health 1987; 8:253. | |||
#Reaven Reaven G., Tsao P.S. “Insulin resistance and compensatory hyperinsulinemia: the key player between cigarette smoking and cardiovascular disease?” J Am Coll Cardiol 2003; 41:1044. | |||
#Ridker Ridker P.M., Cushman M., Stampfer M.J., et al. “Inflammation, aspirin, and the risk of cardiovascular disease in apparently healthy men.” N Engl J Med 1997; 336:973. | |||
#Ridker2 Ridker P.M., Glynn R.J., Hennekens C.H. “C-reactive protein adds to the predictive value of total and HDL cholesterol in determining risk of first myocardial infarction.” Circulation 1998; 97:2007. | |||
#Rimm Rimm E.B., Ascherio A., Giovannucci E., et al. “Vegetable, fruit, and cereal fiber intake and risk of coronary heart disease among men.” JAMA 1996; 275:447. | |||
#Rolland Rolland P.H., Friggi A., Barlatier A., et al. “Hyperhomocysteinemia-induced vascular damage in the minipig. Captopril-hydrochlorothiazide combination prevents elastic alterations.” Circulation 1995; 91:1161. | |||
#Ronksley Ronksley P.E., Brien S.E., Turner B.J., et al. “Association of alcohol consumption with selected cardiovascular disease outcomes: a systematic review and meta-analysis.” BMJ 2011; 342:d671. | |||
#Rose Rose G., Hamilton P.J., Colwell L., Shipley M.J. “A randomised controlled trial of anti-smoking advice: 10-year results.” J Epidemiol Community Health 1982; 36:102. | |||
#Rossouw Rossouw J.E., Anderson G.L., Prentice R.L., et al. “Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results From the Women's Health Initiative randomized controlled trial.” JAMA 2002; 288:321. | |||
#Rozanski Rozanski A., Blumenthal J.A., Kaplan J. “Impact of psychological factors on the pathogenesis of cardiovascular disease and implications for therapy.” Circulation 1999; 99:2192. | |||
#Rubin Rubin R., Strayer D.S., Rubin E. (ed.). (2008). Rubin's Pathology: Clinicopathologic Foundations of Medicine Fifth Edition. Baltimore: Lippincott Williams & Wilkins, p387-408 | |||
#Sacks Sacks F.M., Pfeffer M.A., Moye L.A., Rouleau J.L., Rutherford J.D., Cole T.G., Brown L., Warnica J.W., Arnold J.M., Wun C.C., Davis B.R., Braunwald E. "The effect of pravastatin on coronary events after myocardial infarction in patients with average cholesterol levels. Cholesterol and Recurrent Events Trial investigators." N Engl J Med. 1996;335(14):1001. | |||
#Shepherd Shepherd J., Cobbe S.M., Ford I., Isles C.G., Lorimer A.R., MacFarlane P.W., McKillop J.H., Packard C.J. "Prevention of coronary heart disease with pravastatin in men with hypercholesterolemia. West of Scotland Coronary Prevention Study Group." N Engl J Med. 1995;333(20):1301. | |||
#Stefanadis Stefanadis C., Tsiamis E., Vlachopoulos C., et al. “Unfavorable effect of smoking on the elastic properties of the human aorta.” Circulation 1997; 95:31. | |||
#Tall Tall, A. R. "CETP inhibitors to increase HDL cholesterol levels." N.Engl.J.Med. 356.13 (2007): 1364-66. | |||
#DHHS The health benefits of smoking cessation. A report of the Surgeon General. DHHS Publication (CDC) 90-8416; U.S. Department of Health and Human Services, Washington, DC, 1990. | |||
#Theorell Theorell, T., Lind, E., Floderus, B. “The relationship of disturbing life-changes and emotions to the development of myocardial infarction and other serious diseases.” Int J Epidemiol 1975; 4:281. | |||
#Vita Vita J.A., Keaney J.F. Jr. “Endothelial function: a barometer for cardiovascular risk? Circulation” 2002; 106:640. | |||
#Estruch pmid=23944307 | |||
</biblio> | |||
Latest revision as of 20:24, 4 March 2024
Ronak Delewi, MD; Hayang Yang, MsC; John Kastelein, MD, PhD
A 53 years old man, without medical history or medication visits the family physician and makes an anxious impression. His friend has recently suffered from a myocardial infarction (MI) and he is worried that he might also soon face the same situation. As for family medical history, he has a father with hypertension and an uncle with diabetes mellitus. He does not seem to have any symptoms or complaints at this moment, but he has been smoking for 25 years and is overweight. Because of these characteristics he is worried that he will suffer from a MI. Upon physical examination, his BMI was 29 kg/m2, RR was 152/90 mmHg and heart rate was 75 bpm. The family physician orders a blood test for lipid profile and glucose. Both turn out to be in the normal range.
The family physician gives the patient advice concerning primary prevention for atherosclerosis; quit smoking, try to achieve weight reduction, do regular physical activity, restrict alcohol consumption to less than 3 drinks a day and follow a varied and balanced diet. Regarding hypertension, the advice is to keep his RR under 140/90 mmHg. Antihypertensive medication is not indicated at this moment, because his 10-years risk of death due to cardiovascular disease (Systematic Coronary Risk Evaluation) is lower than 20%. He is advised to undergo regular checkups of cardiovascular risk profile or report to the doctor’s office in case of chest pain.
Introduction
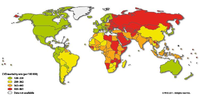
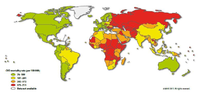
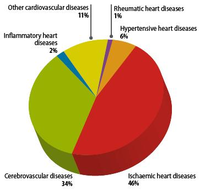
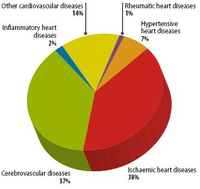
Since the 20th century, cardiovascular disease (CVD’s) has grown to be the leading cause of death and disability in the world, illustrated by 17.3 million deaths per year in 2008. Amongst cardiovascular disease, coronary heart disease (46% among males, 38% among females) and cerebrovascular disease (34% among males, 37% among females) account for the largest proportion of CVD. In 2008, heart attack and stroke were responsible for 7.3 million deaths and 6.2 million deaths, respectively. Obstructive coronary and cerebrovascular diseases are caused, in the vast majority of cases, by atherosclerosis. Atherosclerotis vascular disease begins early in life and over time can eventually lead to obstructive arterial disease. Once atherosclerotic lesions become clinically significant, serious acute complications such as ischemic heart disease, MI and stroke may occur. This chapter deals with the complex pathological process of atherosclerosis, possible consequences of atherosclerosis and the most recent treatment for atherosclerosis in order to prevent CVD’s.
Arterial vessel in homeostasis
The core of the pathogenesis of atherosclerosis is a disease state of the arterial wall. In order to understand the pathogenesis of atherosclerosis, it is thus necessary to know about the function and normal morphology of non-pathological arteries.
Three layers of arterial vessel
The normal arterial vessel consists of 3 layers, namely intima, media and outer adventitia.
The intima is located closest to the arterial lumen and is therefore most ‘intimate’ with the blood. This layer is composed of a single layer of endothelial cells (endothelium), connective tissue, and several smooth muscle cells. The endothelium functions as an active metabolic barrier as well as a carrier between blood and the arterial wall. It plays a crucial role in atherosclerosis. Connective tissue consists of a matrix of collagen, proteoglycans and elastin. Lymphocytes, macrophages and other types of inflammatory cells may occasionally reside in the intima.
The media is the middle layer and its inner and outer boundaries are formed by the internal and external elastic laminae. The media consists of layers of smooth muscle cells with contractile and synthetic function. As for the contractile function, smooth muscle cells enable vasoconstriction and vasodilatation. As for the synthetic function, they are responsible for the growth of the vascular extracellular matrix.
The most external vessel wall layer is called the adventitia and contains fibroblasts, connective tissue, nerves, lymphatics and vasa vasorum. Inflammatory cells may also occasionally reside in the adventitia.
There is a constant dynamic interchange between the arterial wall and its cellular components and the surrounding extracellular matrix. By understanding the physiology of this dynamic interchange and the function of each cellular component, the dysfunction of these cellular components leading to atherogenesis can be better understood.
Cellular components involved in atherosclerosis
Endothelial cells
The normal artery wall contains endothelial cells that manage the homeostasis of the wall by structural, metabolic, and signaling functions. The endothelium plays a role as a barrier to elements contained in the blood, but is also an active biologic interface between the blood and other tissues, regulating cellular and nutrient trafficking. It has several important functions such as keeping certain elements in blood separated from the vessel and maintaining a balance between pro-coagulant and anticoagulant activity, pro- and anti-inflammatory response, and contracted and relaxed vasomotor tone.
The endothelium produces antithrombotic molecules in order to prevent blood from clotting. Certain molecules such as heparin sulfate, thrombomodulin, and plasminogen rest on the endothelial surface whereas molecules such as prostacyclin and nitric oxide (NO) enter the blood. Endothelium can produce prothrombotic molecules when it encounters stressors; however, it normally maintains a balanced anticoagulant state, maintaining blood fluidity.
Endothelial cells also have an important function as a regulator of the immune response. In a normal situation without pathologic stimuli, endothelial cells are not capable to attract and bind patrolling leukocytes, thus maintaining an anti-inflammatory state. When local injury or infection initiates pathologic stimulation, endothelial cells respond by secreting chemokines that attract white blood cells to the injured area. Additionally, endothelium produces cell surface adhesion molecules, which recruit mononuclear cells to the endothelium and therefore promote their migration to the injury site. This response is important for the development of atherosclerosis.
Another function of endothelium is to modulate contraction of smooth muscle cells in the media by releasing substances such as vasodilators and vasoconstrictors. Vasodilators (e.g. NO, prostacyclin) and vasoconstrictors (e.g. endothelin) fine-tune the resistance of the vessel and subsequently alter the arterial blood flow. Endothelium normally maintains a state of net relaxed vasomotor tone with a predominance of vasodilators. Endothelium can also respond to various physical stimuli such as shear stress and can additionally dilate the blood vessel. The endothelium principally regulates such response through release of NO. This endothelial-dependent response is called flow-mediated vasodilation (FMD), which can be measured for clinical evaluation of endothelial function. For example, impairment of FMD is observed in the early stages of atherosclerosis. However, endothelial function tests are currently not recommended to be used for surrogate markers in clinical practice since the tests are technically challenging and the validation of clinical benefits in the evaluation of cardiovascular risk requires more evidence.
As mentioned earlier, endothelial cells can respond to or in other words get ‘activated’ due to changes in the local extracellular milieu. Examples of such changes are common stresses (e.g. shear stress and mild changes in temperature), transient infections and minor trauma. The term ‘endothelial cell activation’ (EC activation) refers to a change from the normal state, illustrated by loss of barrier function, pro-adhesive (leukocyte adhesion), vasoconstriction, and procoagulant properties. EC activation is not necessarily linked to disease and can be temporary and mild or permanent and severe.
In conclusion, the normal arterial endothelium consists of a dynamic interface with net anticoagulant properties, net relaxation of smooth muscle cells and anti-inflammatory characteristics. Endothelial cells may react to various changes in homeostasis and become ‘activated endothelial cells’.
Vascular smooth muscle cells
As mentioned earlier, smooth muscle cells have two functions, namely contractile and synthetic. Vasoconstriction and vasodilatation are regulated by various vasoactive substances such as angiotensin II, acetylcholine, NO and endothelin, which are released by endothelium. Another element of contractile function is the elasticity of the vessel, which is regulated by the lamina elastica. They are situated between the smooth muscle cells and are responsible for the stretching of the vessel during systole and diastole. This function is crucial in the pathogenesis of atherosclerosis, because it prevents the weakening of the vessel wall that can prevail as a complication of atherosclerosis. For example, aneurysm due to weakening of the vessel wall is a serious complication of atherosclerosis.
It is important to understand the synthetic function of smooth muscle cells since the dysfunction of it is thought to contribute to the pathogenesis of atherosclerosis. Normally the smooth muscle cells synthesize collagen, elastin and proteoglycans that form the connective tissue matrix of the vessel wall. Smooth muscle cells can also synthesize vasoactive and inflammatory mediators such as interleukin-6 (IL-6) and tumor necrosis factor-α (TNF- α). These mediators stimulate leukocyte migration and induce the endothelial cells to express leukocyte adhesion molecules as mentioned earlier. This synthetic function is found to be more dominant in case of an atherosclerotic plaque, which is illustrated in the next section (1.2). Although smooth muscle cells rarely divide in normal circumstances, it can proliferate in response to injury, which is an important sign of atherosclerotic plaque formation.
Extracellular matrix
Vascular extracellular matrix in the media consists of elastin, proteoglycans and fibrillar collagen, which are principally synthesized by smooth muscle cells as mentioned earlier. With the provision of flexibility by elastin and biomechanical strength by fibrillar collagen, the arterial vessel is able to maintain the structural integrity despite high pressure within the lumen.
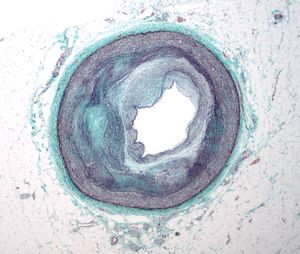
1.2 Arterial vessel with atherosclerosis
Three pathologic stages of atherogenesis
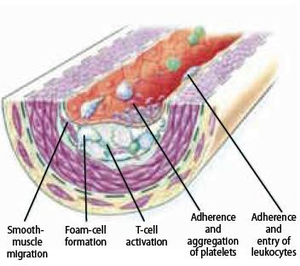
Atherogenesis can be divided into five key steps, which are 1) endothelial dysfunction, 2) formation of lipid layer or fatty streak within the intima, 3) migration of leukocytes and smooth muscle cells into the vessel wall, 4) foam cell formation and 5) degradation of extracellular matrix. Via these consecutive steps, an atherosclerotic plaque is formed. The formation of the plaque can also be divided into three major stages namely 1) the fatty streak, which represents the initiation 2) plaque progression, which represents adaption and 3) plaque disruption, which represents the clinical complication of atherosclerosis.
Initiation and formation of atherosclerotic plaque
The earliest visible signs of atherogenesis are the fatty streak and pre-existing lesions of adaptive intimal thickening. Fatty streak is a yellow discoloration on the surface of the artery lumen, which is flat or slightly elevated in the intima and contains accumulations of intracellular and extracellular lipid. At this stage of initiation, the fatty streak doesn’t protrude substantially into the artery wall nor impedes blood flow. This process is already visible in most people by the age of 20. At this stage, there are no symptoms and this lesion may even diminish over time. Initiation of fatty streak development is most likely caused by endothelial dysfunction, since it involves entry and modification of lipids within the subintima. This modified layer of lipids creates a proinflammatory environment and initiates the migration of leukocytes and formation of foam cells (Figure 5). Intimal thickening mainly contains smooth muscle cells and proteoglycan-collagen matrix with a few or no infiltrating inflammatory cells.
Endothelial dysfunction
| Table 6. Factors correlated with endothelial dysfunction |
|---|
|
Endothelial dysfunction is a primary event in atherogenesis, which can be caused by various agents, such as physical stress and chemical irritants. Endothelial dysfunction is also observed in other pathological conditions, which are often related to atherosclerosis such as hypercholesterolemia, diabetes, hypertension, heart failure, cigarette smoking and aging.
Endothelial cells can display different reactions according to various levels of physical stress. There are two atheroprotective endothelial functions from physical stress. When endothelial cells are exposed to laminar flow, which display minimal physical stress, they secrete NO. NO functions as an anti-atherosclerotic substance through vasodilation, inhibition of platelet aggregation and anti-inflammatory effects. The second function is executed, when exposed to laminar flow by an expression of the antioxidant enzyme superoxide dismutase. This enzyme performs anti-atherosclerotic role by acting against reactive oxygen species, which are produced by chemical irritants or transient ischemia in the vessel.
| Table 7. Interventions that enhance endothelial function |
|---|
|
Unfortunately, these two atheroprotective endothelial functions can be impaired by several factors. The first factor is disturbed flow (low shear stress with rapid fluctuations), which is typically located at arterial branch points and bifurcations and can impair the protective functions. This is well illustrated by the difference in prevalence of atherosclerosis between branched arteries and bifurcated vessels. Bifurcation areas such as the common carotid and left coronary arteries are common deposition sites for atherosclerosis than arteries with few branches such as the internal mammary artery. Thus, many observations show that the distribution of atherosclerotic lesions is common in large vessels and they vary in location and frequency among different vascular beds. These findings encourage a belief that hemodynamic factors play an important role in atherogenesis. Furthermore, the fact that hypertension intensifies the severity of atherosclerotic lesions additionally supports this hypothesis.
Another major factor that can impair the atheroprotective endothelial function is chemical irritants such as cigarette smoking, abnormally high circulating lipid levels and high glucose level (diabetes mellitus). They can contribute to endothelial dysfunction and are all well- known risk factors for atherosclerosis. Exposure to chemical irritants promotes endothelial dysfunction by increasing endothelial production of reactive oxygen species, which alter the metabolic and synthetic functions of endothelial cells. As a result, the endothelium become inclined to exhibit proinflammatory processes, such as secreting inflammatory cytokines.
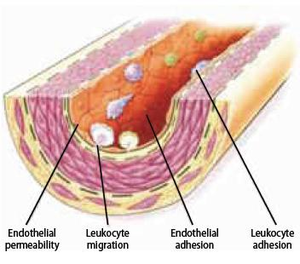
In conclusion, hemodynamic and chemical stressors contribute to disturbance of endothelial homeostasis and promote endothelial dysfunction. This results in impairment of permeability barrier function, secretion of inflammatory cytokines, stimulation of adhesion molecules on the cell surface that promote leukocyte recruitment, and altered antithrombotic properties and release of vasoactive molecules (Figure 8). Consequently, these effects establish the groundwork for further advancement of atherosclerosis.
Lipoprotein entry and modification
Disruption of the integrity of endothelial barrier due to endothelial dysfunction allows the passage of circulating lipoproteins (low-density lipoprotein, LDL) into the intima. By binding to proteoglycans, LDL particles start to accumulate. This accumulation is a critical process in atherogenesis since LDL may undergo chemical modifications while residing longer in the intima. It is needless to say that an elevated circulating LDL concentration strongly contributes to this accumulating process. Another major risk factor for this process is hypertension since it causes augmented vessel wall stress. Elevated vessel wall stress influences smooth muscle cells to synthesize proteoglycans in the intima, promoting LDL-binding with proteoglycans and therefore contributing to “trapping” of lipoproteins and lipid accumulation within the intima. At this point, macrophages adhere to dysfunctional endothelial cells and transmigrate into the intima. These macrophages are called ‘foam cells’ after they have taken up lipids.
As mentioned earlier, chemical modification occurs with LDL when chronic accumulation takes place inside the intima. There are several types of chemical modification that may occur. One is called oxidation and it results from the chemical reaction of reactive oxygen species and pro-oxidant enzymes produced by endothelial or smooth muscle cells, or macrophages penetrating the intima. This type of oxidative stress leads to cellular dysfunction and damage in endothelial cells and macrophages. Furthermore chronic hyperglycemia can stimulate glycation of LDL that may ultimately alter LDL into an antigenic and proinflammatory molecule. This explains why diabetes mellitus is a major risk factor for atherosclerosis. The biochemical modification of LDL into a proinflammatory molecule contributes to the inflammation process established by endothelial dysfunction. Furthermore, the oxidized LDL molecule induces tissue damage, which can initiate angiogenesis, forming new vasa vasorum in the plaque. It also induces leukocyte recruitment and foam cell formation in the fatty streak throughout the plaque development.
Leukocyte recruitment
Leukocyte recruitment to the arterial wall is another key step in atherogenesis, which is dependent on two important factors; expression of leukocyte adhesion molecules (LAM) on the endothelial wall and chemoattractant signals that direct diapedesis (intruding of molecules through the intact vessel wall). These two factors mainly direct monocytes to the atherosclerotic lesion. T lymphocytes that play a central role in the immune system reside within plaques at all stages of atherogenesis, mainly producing cytokines.
As mentioned earlier, modified LDL can maintain leukocyte recruitment by inducing LAM and chemokine expression. It can also stimulate endothelial and smooth muscle cells to produce proinflammatory cytokines. These proinflammatory cytokines can also induce LAM and chemoattractant cytokine expression, equivalent to the working of modified LDL. In conclusion, modified LDL can directly or indirectly promote leukocyte recruitment and atherogenesis.
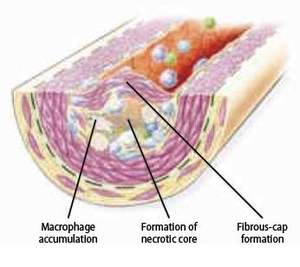
Foam cell formation
When monocytes enter the intima, they differentiate into phagocytic macrophages. These phagocytic macrophages may become foam cells when they absorb lipoproteins. They don’t phagocyte LDL with a classic cell surface LDL-receptor, since it does not recognize modified LDL, but with a family of ‘scavenger’ receptors that do bind and internalize modified LDL. Uptake by scavenger receptors avoids negative feedback inhibition from the high cholesterol content unlike the classic LDL-receptors, and allows the macrophages to imbibe cholesterol-rich lipid that results into the formation of foam cells. This uptake seems to be beneficial at first sight, since it absorbs the inflammatory modified-LDL, however since these foam cells have impaired trafficking, they will be locally accumulated in the plaque and encourage the plaque progression by serving as a source of proinflammatory cytokines.
Plaque progression
The atherosclerotic plaque at this stage is called fibrous cap atheroma featuring two characteristics, which are lipid-rich necrotic core and encapsulation by a fibrous cap (Figure 9). The fibrous cap is an area between the vessel lumen and the core of the plaque, which contains dead foam cells, macrophages, smooth muscle cells, lymphocytes and extracellular matrix. A distinctive hallmark of this phase is necrosis with macrophage infiltration around a lipid pool and loss of proteoglycans or collagen. At this point, the deposition of free cholesterol is not easily visible and the plaque does not always cause luminal restriction of blood flow due to a compensatory outward remodeling of the plaque wall. This remodeling preserves the diameter of the vessel lumen and thus may evade detection by angiography. Continuous plaque growth at a later stage contains cellular debris, higher free cholesterol and results into complete depletion of extracellular matrix. From this stage, the fibrous cap atheroma may go through episodes of hemorrhage with or without calcification and even fibrous cap disruption. Progressive vessel narrowing may result in ischemia and can cause ischemic symptoms such as angina pectoris or intermittent claudication.
Smooth muscle cell migration
Smooth muscle cells play a central role at the phase of transition from fatty streak to plaque formation. During this phase, smooth muscle cells migrate from the media to the intima. After migration, smooth muscle cells proliferate within the intima and secrete extracellular matrix macromolecules. Additionally, foam cells, activated platelets and endothelium stimulate substances that induce the migration and accumulation of smooth muscle cells. For example, foam cells release platelet derived growth factor (PDGF), cytokines and growth factors that directly contribute to the migration and proliferation process, and they also activate smooth muscle cells and leukocytes to reinforce inflammation in the atherosclerotic lesion. Although plaque progression is traditionally known as a gradual and continuous process, recent evidence claims that this process can be strongly accentuated by bursts of smooth muscle replication. The observation of small ruptures within the plaque occurring without any clinical symptoms or signs supports this suggestion. These small ruptures expose tissue factor secreted by foam cells that stimulates coagulation and microthrombus formation in the lesion. Such microthrombi contain activated platelets that release additional factors such as PDGF and heparinase that can further stimulate local smooth muscle cell migration and proliferation. Heparinase stimulates smooth muscle cell migration and proliferation by degrading heparan sulfate, which normally counteracts this process.
Extracellular matrix metabolism
Metabolic processes in extracellular matrix play a central role in bridging the plaque progression to plaque rupture. Ultimately, this process weakens the fibrous cap, predisposing it to rupture. This process is influenced by the balance of matrix deposition synthesis by smooth muscle cells and degradation by matrix metalloproteinases (MMP), a class of proteolytic enzymes. For example, PDGF and TGF-β stimulate interstitial collagen production, while inflammatory cytokines such as IFN-γ inhibits collagen synthesis. TGF-β also induces formation of fibronectin and proteoglycans. It is an important regulator since it enhances the expression of protease inhibitors, leading to the inhibition of proteolytic enzymes that promote matrix degradation. On the other hand, inflammatory cytokines weaken the fibrous cap by stimulating local foam cells to secrete MMP that degrades collagen and elastin of the fibrous cap. Furthermore, the deeper parts of the thickened intima undergo necrosis due to poor nourishment.
Plaque rupture
Integrity of plaque
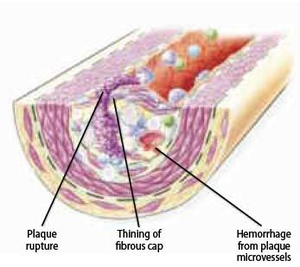
Chronic shifting of the balance towards extracellular matrix metabolism leads to serious consequences for the plaque integrity. As mentioned earlier, it accelerates inflammatory stimulation or activation of apoptosis pathways and therefore leads to death of smooth muscle and foam cells. Cell death leads to release of cellular contents, whereby more lipids and cellular debris is absorbed to the dynamic lipid core. Due to this process, the size of the lipid core grows and as a result alters biomechanical environment and hence the stability of the plaque. One example of this is a plaque border adjacent to the normal tissue, called shoulder region, which is the main location where the hemodynamic stress is focused. As the size and the protrusion of the plaque in the vessel increase, the hemodynamic stress will also increase around the shoulder region. Furthermore, local accumulation of foam cells and lymphocytes at this site makes the plaque more susceptible to rupture by accelerating degradation of extracellular matrix. However, although shoulder area is considered as the weakest point where the fibrous cap would mostly likely rupture, there have been autopsy studies that showed an equal number of ruptures occurring at the midportion of the fibrous cap. When the fibrous cap is very thick and contains small lipid core, the plaque is called stable and it may reinforce the narrowing of the artery, but on the other hand diminishes the susceptibility to rupture. Plaques with thinner fibrous caps are called vulnerable plaques. They are identified by a large necrotic core, rich with lipid, taking about 25% of the plaque area, and a thin fibrous cap of less than 65 µM thickness, which separates the necrotic core from the vessel lumen. Vulnerable plaque is infiltrated by a large amount of macrophages and a smaller amount of T-lymphocytes. It typically lacks smooth muscle cells due to apoptosis. This type of lesion causes less obstruction in the artery, but is more fragile and has higher susceptibility to rupture and trigger thrombosis than a thick fibrous cap. At this stage, plaque hemorrhage can occur due to rupture of vasa vasorum within a plaque. Vasa vasorum is a newly formed vascularization in the plaque due to tissue damage. Due to its fragility it may rupture easily, increasing the risk to form intraplaque hemorrhage. Intraplaque hemorrhage may lead to subsequent rupture of the fibrous cap (Figure 10) or occlusion of the vessel through intramural hematoma. Plaque calcification is another factor that contributes to plaque rupture. It usually occurs in areas of necrosis and elsewhere in the plaque and can eventually lead to higher rigidity of the vessel wall. Calcification is dependent on mineral deposition and resorption by osteoblast-like and osteoclast-like cells in the vessel wall. In conclusion, there are seven important factors associated with plaque ruptures; range of inflammation area, considerable size of lipid core, fibrous cap thinner than 65 µM, apoptosis leading to fewer smooth muscle cells, disrupted balance of proteolytic enzymes and their inhibitors, plaque calcification, and hemorrhage in the plaque. Although it remains difficult to foresee the clinical consequences, progression to a complicated plaque can lead to major cardiovascular disease, mostly affecting individuals in their 60s and 70s, although it may also occur among people at an earlier age.
Thrombogenic potential after rupture
When the fibrous cap is ruptured, the highly thrombogenic components of the necrotic core, including tissue factor, gets in direct contact with the circulating monocytes in the blood. It is believed that these circulating monocytes in the blood play a stronger role as a source of tissue factor than the necrotic core. Tissue factor stimulates platelet activation and thus can initiate and propagate thrombus. The thrombus formed at the rupture site is called white thrombus due to its grossly white appearance of rich platelet. At the proximal and distal ends near the site of white thrombosis there is another type of thrombus composed of layers of red blood cells and fibrin and is therefore called red thrombus. Thrombosis can be healed through several processes such as penetration of smooth muscle cells, neovascularization via vasa vasorum, proliferation of extracellular matrix, inflammation and re-endothelialization on the luminal surface. Thus clinically, ruptures can be silent and heal, without major clinical complications such as MI and stroke. For example, small non-occlusive thrombi may be reabsorbed into the plaque, continuing the process of smooth cell growth and fibrous deposition. The extent of how occlusive and transient the thrombus will be is largely dependent on the thrombogenic potential of the plaque.
The counter-balancing of coagulation and fibrinolysis also determines the probability of a major clinical event due to occlusive thrombosis. Inflammatory stimuli in the plaque environment incite smooth muscle cells, endothelial cells, and foam cells to release tissue factor that initiates the extrinsic coagulation pathway. Inflammatory stimuli also stimulate expression of antifibrinolytics such as plasminogen activator inhibitor-1 and consequently enhance thrombosis. As mentioned earlier, the activated endothelial cells also contribute to thrombosis and coagulation by depositing fibrin at the vascular wall. Thus, the inflammatory and dysfunctional condition of the plaque environment decreases the counterbalancing of coagulation and fibrinolysis, increasing the probability of major clinical complications of atherosclerosis.
The concept of ‘vulnerable plaque’ has developed into a new concept of ‘vulnerable patient’ as the concept of pathogenesis of atherosclerosis was linked to a person’s susceptibility to coagulation and thus vascular events, which can be influenced by many personal factors such as genetics (e.g. procoagulant prothombin gene mutation), coexisting condition (e.g. diabetes), and lifestyle factors (e.g. smoking, obesity).
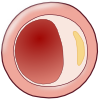
|
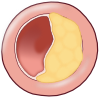
|

|
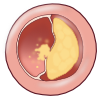 |
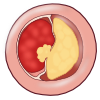
|
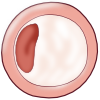
| ||||
| Progression of coronary atherosclerosis can be gradual (bottom) or can lead to plaque rupture with acute occlusion of a coronary vessel due to clot formation | ||||
Complications of atherosclerosis
| Figure 11. Most common locations of atherosclerosis |
|---|
|
The clinical complications of atherosclerosis are highly dependent on the location and size of affected vessels, the duration of the chronic process, and the type of plaque, since the severity of impairment of atherosclerosis differs throughout the vasculature. For example, ‘stable plaque’ can easily result into angina pectoris due to its thick fibrous cap that directly affects the diameter of the relatively small coronary vessels. On the other hand, ‘vulnerable plaque’ is non-stenotic, but can easily cause acute thrombosis and therefore myocardial infarction due to its fragility towards rupture when located at physically stressed areas such as bifurcations. Often with ‘vulnerable plaques’ there are relatively few symptoms, however they are more numerous and dispersed throughout the arteries compared to ‘stable plaque’. Thus, you can either have an occlusion due to the growing plaque or due to the embolization of the ruptured fragments of the original plaque. Due to the difficult detection of ‘vulnerable plaques’ while they are widely dispersed, it is highly important to tackle the risk factors prior to plaque rupture. Thus in the following paragraph, we will highlight the clinical risk factors associated with atherosclerosis. The four major clinical consequences of atherosclerosis are listed and explained below.
- Acute narrowing of the vessel lumen: When the plaque ruptures, it will release its pro-coagulants in the bloodstream and that will lead to the formation of thrombus at the rupture site. The rupture often occurs at sites of erosion and fissuring on the fibrous cap surface. This thrombus may cause a complete occlusion of a particular vessel and result in ischemic necrosis (infarction) of the tissue that this particular vessel is supplying to. Clinically this is manifested as stroke, MI, gangrene of several possible organs such as intestine, spleen or lower extremities. These occlusions may also dissolve spontaneously due to pro-fibrinolytic enzymes such as streptokinase and tissue plasminogen activator (TPA).
- Chronic occlusion: When the occlusion is gradual and incomplete, it may chronically disturb the blood supply to tissues in the distribution of the affected vessel. This can result in chronic ischemia of those tissues that can additionally lead to complaints of angina pectoris or intermittent claudication or to organ atrophy (e.g. atrophy of kidney, intestines and skin due to impairment of blood flow in renal artery, mesenteric artery, peripheral vasculature among diabetics.).
- Embolism: Embolization is the transfer of the fragments of disrupted atheroma to distal vascular sites, which results into occlusion of those sites. For example, fragments of thrombi in abdominal aorta may transfer to the popliteal artery subsequently resulting in gangrene of the leg. Ulceration of atheroma may also produce ‘cholesterol crystal emboli’. This type of emboli is visualized as needle-shaped areas in affected tissues, mostly detected in the kidney.
- Aneurysm: After a chronic period, atherosclerotic lesion may extend into the medial layer, resulting into atrophy and loss of elastic tissue. This can subsequently cause dilatation and weakness of the artery, forming aneurysm. Over time, aneurysms may suddenly rupture and result in a life-threatening situation for the patients.
Risk factors of atherosclerosis
| Figure 13. Nine modifiable risk factors for atherosclerosis according to INTERHEART study |
|---|
|
Recent studies have shown that atherosclerosis is not just the inevitable process of aging, but also a process with many modifiable components. A worldwide INTERHEART study has established the importance of nine potentially modifiable risk factors for atherosclerosis, which account for over 90% of the population-attributable risk of a first MI (figure 12). A variety of non-modifiable risk factors such as advanced age, gender and hereditary coronary heart disease are important to recognize in patients with atherosclerosis. Recently the role of several biological markers associated with the development of cardiovascular events is accentuated since one out of five cardiovascular events occurs in patients lacking the earlier mentioned risk factors.
Common risk factors
Dyslipidemia
One of the major modifiable risk factors for atherosclerosis is hypercholesterolemia. Studies show that dyslipidemia (defined as an elevated apo B to apo A-1 ratio) was responsible for 49% of the population-attributable risk of a first MI. In countries with high consumption of saturated fat and high cholesterol levels (e.g. the United States), observational studies have shown that the mortality rates from coronary disease are higher compared with countries with traditionally low consumption of saturated fat and cholesterol levels (e.g. Japan). Several trials have shown that the risk of ischemic heart disease positively correlates with higher total serum cholesterol levels. For example, the impact of hypercholesterolemia can be illustrated by an observational result from the Framingham Heart Study, which shows that a person with a total cholesterol level of 240 mg/dl has twice the coronary risk a person would have with a cholesterol level of 200 mg/dl. However, it is a mistake to think that all lipoproteins consisting of cholesterol are harmful since cholesterol can provide critical functions to all cells that need to form membranes and to synthesize products such as steroid hormones and bile salts.
| Figure 14. Recommendations regarding dyslipidemia |
|---|
General recommendations:
Specific diet recommendations:
|
Incidence of atherosclerosis and coronary artery disease increases with higher levels of LDL particles. As mentioned earlier, LDL can accumulate in the intima of the artery in excess and undergo chemical modifications that activate endothelial cells to proceed to atherosclerosis. When people generally refer to ‘bad cholesterol’, they are referring to LDL particles. On the other hand, high levels of high-density lipoprotein (HDL) constitute ‘good cholesterol’ since it protects against atherosclerosis by reversing the cholesterol transport from peripheral tissues to the liver for disposal and functions as an antioxidant. In order to give additional explanation to what is ‘bad cholesterol,’ all lipid and lipoprotein abnormalities that are associated with higher coronary risk will be named subsequently: increased total cholesterol, increased LDL-cholesterol, low HDL-cholesterol, elevated total-to-HDL-cholesterol ratio, hypertriglyceridemia, increased non-HDL-cholesterol, elevated lipoprotein A, elevated apolipoprotein B (apo B is primarily found in LDL), decreased apolipoprotein A-I (apo A-1 is found in HDL), small and dense LDL particles.
There are several causes for persistent elevated levels of LDL, such as high fat consumption or genetic abnormalities (e.g. familial hypercholesterolemia). Familial hypercholesterolemia is a condition with deficient LDL receptors that cannot efficiently dispose LDL from the circulation. There are two types of this disease with different manifestations. Patients with the heterozygote type have only one defective gene for the receptor and suffer from high serum level of LDL, easily developing atherosclerosis. Homozygotes have a complete lack of normal LDL receptors and thus may experience cardiovascular events already in the first decade of life. In the absence of genetic abnormalities, the quantity of cholesterol in serum is strongly related to saturated fat consumption.
Lipid-Altering therapy
Controlling serum lipids is a key step to limit the consequences of atherosclerosis. Major clinical trials have shown that coronary events and mortality significantly decreased when total and LDL-cholesterol levels were reduced for the primary and secondary prevention of CAD events.
One of the most important strategies to reduce the complications of atherosclerosis is diet and exercise. In order to decrease cholesterol, Mediterranean diet is often recommended. Mediterranean diet consists of low animal fat, high olive oil, moderate energy consumption, nuts, vegetables, regular and moderate wine, lots of whole grains and beans. A meta-analysis of six randomized trials showed that Mediterranean diet led to greater reduction in total cholesterol than low fat diets among overweight/obese population. Mediterranean-styled diet in this context means replacement of saturated fats with polyunsaturated fats such as omega-3 fatty acid and α-linolenic acid. Polyunsaturated fats are potential anti-atherogenic due to their inhibiting action on cytokine-induced expression of leukocyte adhesion molecules at endothelial cells. Exercise and loss of excessive weight also contributes to improve abnormal lipid levels by reducing triglycerides and increasing HDL.
In primary prevention, pharmacologic agents are the second option when lifestyle modifications fail to achieve targeted lipid profile. There are several groups of lipid-altering medicines such as HMG-CoA reductase inhibitors (statins), niacin, fibric acid derivatives, cholesterol intestinal absorption inhibitors, and bile acid-binding agents. In the clinical setting, statins are widely used, being the most cost-effective LDL-lowering drugs. They reduce intracellular cholesterol concentration by inhibiting HMG-CoA reductase, which is an enzyme that synthesizes cholesterol. This results into increased LDL-receptor expression and therefore leads to higher clearance of LDL molecules from blood. They also affect the liver and thereby lower the rate of VLDL synthesis, which results into lower levels of serum triglyceride. Statins also raise HDL, but this mechanism is not fully understood yet.
Large studies, which have evaluated the effects of statin therapy, showed that ischemic cardiac events, the occurrence of MI and mortality rates were significantly reduced by implementing statin therapy. This significant improvement did not only apply to people with known preexisting atherosclerotic disease, but also to people within lower ranges of LDL and without preexisting atherosclerotic disease. For example, West of Scotland Coronary Prevention Study (WOSCPS trial) evaluated the effect of pravastatin on rates of nonfatal MI or CHD death, nonfatal MI, all cardiovascular deaths and total mortality among patients with hypercholesterolemia without preexisting CVD for five years. Use of pravastatin resulted in 31% risk reduction (p<0.001) in nonfatal MI or CHD death, and a 32% risk reduction (p=0.033) in all cardiovascular deaths as compared to the control group.
Inhibiting HMG-CoA reductase results in several mechanisms that explain the beneficial effect of using statins. One beneficial mechanism is via lowering LDL and raising HDL. This results into less lipid content in atherosclerotic plaques and improve their biologic activity. Furthermore, the anti-thrombotic and anti-inflammatory profile is enhanced by other mechanisms such as increased NO synthesis and fibrinolytic activity, inhibition of smooth muscle proliferation and monocyte recruitment, and reduced production of matrix-degrading enzymes by macrophages. Several studies suggest that other mechanisms also contribute to the anti-inflammatory profile. For example, statins reduce endothelial expression of leukocyte adhesion molecules and macrophage tissue factor production by inhibiting the macrophage cytokines or by activating PPAR-α. Another anti-inflammatory action of statins, supported by clinical trials is reducing the serum level of C-reactive protein, which is a marker of inflammation.
Despite the effectiveness of statins, niacin, fibric acid derivatives, cholesterol intestinal absorption inhibitors, and bile acid-binding agents in managing serum lipid levels, further advancements in pharmacologic lipid management have introduced additional options for reducing cardiovascular risk associated with atherosclerosis. Two notable additions to the lipid-lowering arsenal are PCSK9 inhibitors and bempedoic acid.
Proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors are a novel class of medications that significantly lower low-density lipoprotein cholesterol (LDL-C). PCSK9 is a protein that binds to LDL receptors on the liver surface, leading to their degradation and preventing them from removing LDL cholesterol from the blood. By inhibiting PCSK9, these drugs increase the number of LDL receptors available to clear LDL from the blood, thus lowering LDL-C levels.
Clinical trials have demonstrated that PCSK9 inhibitors, such as evolocumab and alirocumab, can reduce LDL-C levels by up to 60% when used alone or in combination with statins. This significant reduction in LDL-C levels has been associated with a decreased risk of cardiovascular events, making PCSK9 inhibitors an important option for patients who are statin-intolerant or for whom statins alone are insufficient to achieve LDL-C targets.
Bempedoic acid represents another innovative approach to lowering LDL-C levels. It acts by inhibiting ATP citrate lyase, an enzyme upstream of HMG-CoA reductase in the cholesterol biosynthesis pathway. This inhibition leads to reduced hepatic cholesterol synthesis and upregulation of LDL receptors, resulting in decreased LDL-C levels. Bempedoic acid has the advantage of being metabolized in the liver, sparing muscle tissue and potentially offering a safer alternative for patients who experience muscle-related side effects with statins.
In clinical trials, bempedoic acid has been shown to lower LDL-C levels by up to 20% when used as monotherapy and has shown additional LDL-C lowering when used in combination with statins. Furthermore, bempedoic acid has been associated with reductions in biomarkers of inflammation, such as high-sensitivity C-reactive protein (hsCRP), suggesting potential anti-inflammatory benefits beyond its lipid-lowering effects.
Tobacco smoking
Tobacco use, including environmental smoking exposure, is known to increase the risk of atherosclerosis and ischemic heart disease based on numerous studies. For example, INTERHEART study shows that smoking is responsible for 36% of the population-attributable risk of a first MI. Other studies showed that smoking is an independent major risk factor for coronary heart disease, cerebrovascular disease and total atherosclerotic cardiovascular disease. The Atherosclerosis Risk in Communities Study measured the direct effect of smoking on the development of atherosclerosis. They measured intima-medial thickness of the carotid artery of 10,914 patients for three years with ultrasound. Their result showed that current smokers had a 50% increased progression of atherosclerosis in comparison to nonsmokers during the study period. Also patients with environmental tobacco smoke exposure (passive smokers) had 20% higher rate of atherosclerotic progress versus patients without environmental smoke exposure.
Tobacco smoking can lead to many mechanisms that contribute to atherosclerosis. Smoking also leads to increased LDL levels, decreased HDL levels in blood and elevated insulin resistance. In addition it enhances oxidative modification of LDL by releasing free radicals and reduces generation of nitric oxide. This can promote endothelial dysfunction and thus lead to impairment of vasodilatation of coronary arteries and reduction of coronary flow reserve even in passive smokers. Tobacco smoking inappropriately stimulates sympathetic nervous system, increasing heart rate, blood pressure and perhaps coronary vasoconstriction. Smoking promotes a prothrombotic environment through inhibition of endothelial release of tissue plasminogen activator, elevation of fibrinogen concentration in blood, enhancement of platelet activity (possibility related to sympathetic activation) and enhanced expression of tissue factor. Smoking can even damage the vessel wall and ultimately cause a decrease in the elasticity of the artery, enhancing the stiffness of vessel wall. Smoking has been associated with increased C-reactive protein and fibrinogen, suggesting a correlation with inflammatory response, which is an important part of atherogenesis. There have also been findings that show higher expression of leukocyte adhesion molecules among smokers than nonsmokers. Smoking may additionally induce tissue hypoxia through displacement of oxygen with carbon monoxide in hemoglobin.
To stop smoking is known as one of the most effective preventive measures of CVD and their complications. Soon after cessation, cardiac risks due to smoking decreases in a short period, and continues to diminish when cessation is permanently preserved. The risk for cardiovascular disease among patients with coronary heart disease decreases 7-47%. Not only does cessation of smoking reduce risk of CVD, but also substantially reduce the risk of all-cause mortality.
Lack of physical activity
| Physical Activity | |
|---|---|
| The following activities have similar benefits to health: | |
| Activity | Duration |
| Washing and waxing a car | 45-60 minutes |
| Washing windows or floors | 45-60 minutes |
| Playing volleyball | 45 minutes |
| Wheeling self in wheelchair | 30-40 minutes |
| Bicycling - 8 km | 30 minutes |
| Pushing a pushchair - 2.5 km | 30 minutes |
| Walking - 3 km | 30 minutes |
| Swimming laps | 20 minutes |
| Playing basketball | 15-20 minutes |
INTERHEART study showed that lack of exercise accounted for 12% of the population-attributable risk of a first MI. Recent evidence shows that physical activity of even a moderate degree can protect against coronary heart disease and all-cause mortality .The beneficial effects of physical exercise are a decrease of triglyceride levels and blood pressure, elevation of HDL, enhancement of insulin sensitivity and production of NO by the endothelial cells, and of course weight loss. Although large scale randomized primary prevention trials are lacking, physical activity should be promoted to anyone with risk of developing atherosclerosis.
Obesity
The American Heart Association has published an article, identifying obesity as an independent risk factor for coronary heart disease. Obesity is correlated with several risk factors for atherosclerosis such as hypertension, insulin resistance, glucose intolerance, decreased HDL serum level and hypertriglyceridemia. Weight loss is an important treatment to prevent many obesity-related risk factors for atherosclerosis that has just been mentioned.
Diet
A healthy diet reduces CVD risk. In general, when following the rules for a healthy diet, no dietary supplements are needed. N-3 polyunsaturated fatty acid (PUFA) consumption mainly from oily fish, is potentially associated with beneficial effects on cardiac risk factors, notably reduction in triglycerides but not all randomized, controlled trials have shown reductions in CV events Thus current recommendations are to increase PUFA intake through fish consumption, rather than from supplements. Recently, the largest study ever conducted with a so-called ‘Mediterranean’ diet, supplemented with extra-virgin olive oil or nuts, reduced the incidence of major cardiovascular events in patients at high risk of CV events but without prior CV disease.[1]
Alcohol consumption
Alcohol is harmful when used chronic or excessive and can lead to various complications such as liver and heart failure, increased cancer risk, neurological complications and injuries. However despite these adverse effects, moderate drinking (US parameters; women: <2 drinks per day, men: <3 drinks per day) may have protective benefits with regards to coronary heart disease according to several prospective cohort studies. These studies showed that moderate drinking resulted in a reduction of risk in coronary heart disease by 40-70% compared to no or heavy drinkers. This beneficial effect was seen in various groups without or with known risk for coronary heart disease and adults older than 65 years old. In a meta-analysis study, alcohol drinkers had lower relative risk for CVD mortality (0.75, 95% CI 0.70-0.80), coronary heart disease mortality (0.75, 0.68-0.81) and incidence of coronary heart disease (0.71, 0.66-0.77) than nondrinkers.
Psychosocial factors
Mentioned by INTERHEART study, psychosocial factors may directly contribute to the early development of atherosclerosis. Psychological stress may directly damage endothelium and indirectly aggravate other common risk factors such as smoking, dyslipidemia and hypertension. Due to the difficulty in quantifying the extent of atherosclerosis, studies showing the relationship between stress and atherosclerosis have been limited. Epidemiologic studies have shown stronger link between psychosocial factors (loss of job, depression and bereavement) and MI and sudden death.
Estrogen Status
Women and men have different risk for cardiovascular diseases throughout life. For example, at young age, men have an estimated four- to fivefold higher risk than women. However this difference diminishes and the age point of no difference is strongly related to the moment of menopause. From this observation, it has been suggested that estrogen may play athero-protective roles, since the levels of estrogen declines after menopause. In premenopausal women, estrogen raises HDL levels and reduces LDL levels in blood. Estrogen can even exhibit antioxidant and antithrombotic properties and can improve endothelium-dependent vasodilatation.
In the past, hormone replacement therapy has been suggested by several studies due to the findings of potential athero-protective roles of estrogen. However, these findings were not confirmed in the randomized primary prevention study of the Women’s Health Initiative not in the HERS trial of secondary prevention. These studies showed that hormone replacement therapy (estrogen-progestin replacement) may increase cardiovascular risk in women and have no cardioprotective effect. Thus hormone replacement therapy is currently not recommended for reducing cardiovascular risk, due to its possible harmfulness consequences according to current clinical trials.
Biomarkers
Biomarkers can serve to identify patients with subclinical atherosclerotic disease that are at risk of developing cardiovascular events.
Homocysteine
Homocysteine is an intermediary amino acid produced during the conversion of methionine to cysteine. A significant positive correlation was found between the serum levels of homocysteine and the incidence of cardiovascular diseases. Although the clear mechanism of this correlation is undetermined, the overall result of the most current evidence suggests that homocysteine can modestly contribute to cardiovascular risk by inducing vascular injury. Homocysteine promotes oxidative stress, intimal thickening, disruption of elastic lamina, hypertrophy of smooth muscle cells, vascular inflammation platelet accumulation and production of occlusive thrombi when elevated in blood. Several conditions can cause hyperhomocystinemia, such as genetic defects in methionine metabolism or insufficient consumption of folic acid, which is involved in the methionine pathway. Such disorders cause premature and severe atherosclerosis. Despite this observational relationship, there is no data yet that proves reducing high serum level of homocysteine will lead to a decrease in atherosclerosis or its complications.
Lipoprotein A
Some studies have concluded that lipoprotein (a) is an independent risk factor for coronary artery disease. As lipoprotein (a) contains apo (a), which structurally resembles plasminogen, lipoprotein (a) interferes with fibrinolysis by competing with plasminogen binding with molecules. This leads to impairment of plasminogen activation, plasmin generation and lysis of fibrin clots. In addition, lipoprotein (a) binds with macrophages through a high-affinity receptor, promoting foam cell production and deposition of cholesterol in atherosclerotic plaques. As with homocysteine, not all studies support this theory of correlation, although increased risk of cardiovascular events appear to correlate with people with highest lipoprotein (a) serum level.
C-Reactive Protein and other markers of inflammation
Since the participation of inflammatory cells and mediators in atherosclerosis is well established, markers of inflammation have received a lot of attention. Several markers of inflammation such as C-reactive protein (CRP), fibrinogen and amyloid A are produced by hepatocytes in an acute phase under the influence of cytokines such as IL-6 when they mobilize from intima to the liver during the fatty streak stage. From these markers, CRP has shown the greatest association with atherosclerosis as a marker of low-grade systemic inflammation. A significant association between elevated CRP level in blood and prevalence of atherosclerosis has been shown in more than 30 epidemiologic studies. Different studies showed that higher basal CRP levels (four-fold higher) were found in patients with MI as compared to controls. Several studies have proposed that elevated plasma CRP can be an independent predictor for many cardiovascular diseases based on the result that CRP plasma value was able to predict the long-term risk of first MI, ischemic stroke or peripheral vascular disease among the male group. In addition, recent studies have shown that CRP also has a role as a mediator in atherogenesis. By inducing adhesion molecule expression and release of IL-6 and monocyte chemoattractant protein-1 via endothelial cells, CRP sustains the inflammatory state of atherosclerosis by recruiting monocytes and lymphocytes.
Infection
A variety of infectious agents such as Chlamydia pneumonia, cytomegalovirus and Helicobacter pylori were identified in the lesions of atherosclerosis and this observation raised the suggestion that these infectious agents may contribute to atherogenesis. However, to date, the definite proof of this theory is lacking and also there haven’t been any clinical studies that showed significant relationship between the antibiotic treatment against these infectious agents and the risk of cardiac events of the survivors of acute coronary syndromes. Chlamydia is a strong candidate among other infectious agents, since they produce heat shock protein 60 (HSP-60) that activates macrophages and stimulates the production of matrix metalloproteinases. Furthermore, HDP-60 can also stimulate foam cell formation, lipoprotein oxidation, and increased pro-coagulant activity, which are the major attributing components of atherosclerosis. Although there is no evidence to date, some researchers believe that exogenous pathogens can cause endothelial injury and inflammation that can lead to initiation or exacerbation of atherosclerosis.
Co-morbidity groups
Hypertension
Hypertension is defined as a systolic blood pressure (SBP) ≥ 140mmHg and/or a diastolic blood pressure (DBP) ≥ 90mmHg. Elevated blood pressure is a well established risk factor for atherosclerosis, including mortality from coronary heart disease and stroke. For example, cardiovascular disease doubles with every 20 mmHg increase in SBP or every 10 mmHg increase in DBP.
One of the mechanisms of hypertension contributing to atherosclerosis is injury of vascular endothelium by elevated hemodynamic stress. Injury of endothelium may increase the permeability of the vessel wall to lipoproteins. Increased blood pressure may also increase the number of scavenger receptors on macrophages, which enhances the development of foam cells. Furthermore, increased cyclic circumferential strain in hypertensive arteries can result into promoting LDL accumulation in the intima and facilitation of their oxidative modification. Finally, hypertension can contribute to atherogenesis due to the presence of Angiotensin II, which not only works as a vasoconstrictor, but also as a pro-inflammatory cytokine.
Antihypertensive therapy
| Figure 16. Lifestyle recommendations for hypertension |
|---|
|
Antihypertensive therapy can either consist of lifestyle interventions or pharmacotherapy. Lifestyle modifications consist of diet, body weight reduction, increased activity, and cessation of smoking. As for diet, high consumption of fruits, vegetables, dairy products low in fat, fish oils, potassium and reduced consumption of sodium and alcohol are recommended.
The indication for pharmacotherapy depends on the severity of hypertension and on the assessment of total CVD risk. Several large trials have shown that pharmacotherapy for hypertension can substantially reduce major cardiovascular events such as MI and stroke. Drug therapy is indicated when chronic SBP ≥ 160mmHg and/or DBP ≥100mmHg, or if target organ damage is present.
Diabetes Mellitus
With estimated global incidence of 170 million people, diabetes mellitus is a large problem worldwide. Diabetes mellitus increases the risk of acute coronary events by three- to five folds and 80% of diabetic patients will face atherosclerosis-related cardiovascular diseases. Risk for atherosclerosis among diabetics is considered to be as high as in patients with previous MI. Based on this observation, the National Cholesterol Education Program report from the United States and guidelines from Europe considers type 2 diabetes to be a CHD equivalent, categorizing it to the highest risk for MI.
There are several possible mechanisms that make this group particularly vulnerable to atherosclerosis. An example of mechanism is non-enzymatic glycation of lipoproteins, which promotes uptake of cholesterol by scavenger macrophages. Furthermore, pro-thrombotic and anti-fibrinolytic properties of diabetes can also contribute to this vulnerability. The high prevalence of endothelial dysfunction among diabetes group leads to reduced bioavailability of NO and enhanced leukocyte adhesion. The most effective prevention of atherosclerosis among diabetes group is tight regulation of serum glucose levels. This intervention significantly reduces the risk of microvascular complications such as retinopathy and nephropathy. Furthermore, intense anti-diabetic regime also reduced macrovascular outcomes such as MI and stroke among a group of diabetes type 1. Additionally managing hypertension and dyslipidemia among diabetic groups also significantly reduces the risk of cardiovascular diseases.
References
- Estruch R, Ros E, and Martínez-González MA. Mediterranean diet for primary prevention of cardiovascular disease. N Engl J Med. 2013 Aug 15;369(7):676-7. DOI:10.1056/NEJMc1306659 |
-
Anderson J.L., Carlquist J.F., Muhlestein J.B., et al. “Evaluation of C-reactive protein, an inflammatory marker, and infectious serology as risk factors for coronary artery disease and myocardial infarction.” J Am Coll Cardiol 1998; 32:35.
-
Barua R.S., Ambrose J.A., Eales-Reynolds L.J., et al. “Dysfunctional endothelial nitric oxide biosynthesis in healthy smokers with impaired endothelium-dependent vasodilatation.” Circulation 2001; 104:1905.
-
Barter P.J., Caulfield M., Eriksson M., et al. “Effects of torcetrapib in patients at high risk for coronary events.” N Engl J Med 2007; 357:2109.
-
Bazzano L.A., He J., Muntner P., et al. “Relationship between cigarette smoking and novel risk factors for cardiovascular disease in the United States.” Ann Intern Med 2003; 138:891.
-
Bennet A., Di Angelantonio E., Erqou S., et al. “Lipoprotein(a) levels and risk of future coronary heart disease: large-scale prospective data.” Arch Intern Med 2008; 168:598.
-
Blood Pressure Lowering Treatment Trialists' Collaboration, Turnbull F., Neal B., et al. “Effects of different regimens to lower blood pressure on major cardiovascular events in older and younger adults: meta-analysis of randomised trials.” BMJ 2008; 336:1121.
-
Burke A.P., Farb A., Malcom G.T., Liang Y., Smialek J.E., Virmani R. "Plaque rupture and sudden death related to exertion in men with coronary artery disease." JAMA. 1999;281(10):921.
-
Camm A.J., Füscher T.F., Serruys P.W., eds. (2010). The ESC Textbook of Cardiovascular Medicine Online Second Edition. Oxford: Oxford University Press.
-
Celermajer D.S., Sorensen K.E., Georgakopoulos D., et al. “Cigarette smoking is associated with dose-related and potentially reversible impairment of endothelium-dependent dilation in healthy young adults.” Circulation 1993; 88:2149.
-
Dauchet L., Amouyel P., Dallongeville J. “Fruit and vegetable consumption and risk of stroke: a meta-analysis of cohort studies.” Neurology 2005; 65:1193.
-
De Backer G., Ambrosioni E., Borch-Johnsen K., et al. “European guidelines on cardiovascular disease prevention in clinical practice: third joint task force of European and other societies on cardiovascular disease prevention in clinical practice (constituted by representatives of eight societies and by invited experts).” Eur J Cardiovasc Prev Rehabil 2003; 10:S1.
-
Downs J.R., Clearfield M., Weis S., Whitney E., Shapiro D.R., Beere P.A., Langendorfer A., Stein EA, Kruyer W, Gotto AM Jr. "Primary prevention of acute coronary events with lovastatin in men and women with average cholesterol levels: results of AFCAPS/TexCAPS. Air Force/Texas Coronary Atherosclerosis Prevention Study." JAMA. 1998;279(20):1615.
-
Eckel R.H., York D.A., Rössner S., et al. “Prevention Conference VII: Obesity, a worldwide epidemic related to heart disease and stroke: executive summary.” Circulation 2004; 110:2968.
-
Emerging Risk Factors Collaboration, Erqou S., Kaptoge S., et al. “Lipoprotein(a) concentration and the risk of coronary heart disease, stroke, and nonvascular mortality.” JAMA 2009; 302:412.
-
Fuchs C.S., Stampfer M.J., Colditz G.A., et al. “Alcohol consumption and mortality among women.” N Engl J Med 1995; 332:1245.
-
Grady D., Herrington D., Bittner V., et al. “Cardiovascular disease outcomes during 6.8 years of hormone therapy: Heart and Estrogen/progestin Replacement Study follow-up (HERS II).” JAMA 2002; 288:49.
-
Harker L.A., Ross R., Slichter S.J., Scott C.R. “Homocystine-induced arteriosclerosis. The role of endothelial cell injury and platelet response in its genesis.” J Clin Invest 1976; 58:731.
-
Haffner S.M., Lehto S., Rönnemaa T., et al. “Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with and without prior myocardial infarction.” N Engl J Med 1998; 339:229.
-
Howard G., Wagenknecht L.E., Burke G.L., Diez-Roux A., Evans G.W., McGovern P., Nieto F.J., Tell G.S. "Cigarette smoking and progression of atherosclerosis: The Atherosclerosis Risk in Communities (ARIC) Study." JAMA. 1998;279(2):119.
-
Jackson R., Scragg R., Beaglehole R. “Alcohol consumption and risk of coronary heart disease.” BMJ 1991; 303:211.
-
Jee S.H., Suh I., Kim I.S., Appel L.J. "Smoking and atherosclerotic cardiovascular disease in men with low levels of serum cholesterol: the Korea Medical Insurance Corporation Study." JAMA. 1999;282(22):2149.
-
Kannel W.B., D'Agostino R.B., Belanger A.J. “Fibrinogen, cigarette smoking, and risk of cardiovascular disease: insights from the Framingham Study.” Am Heart J 1987; 113:1006.
-
Klein S., Burke L.E., Bray G.A., et al. “Clinical implications of obesity with specific focus on cardiovascular disease: a statement for professionals from the American Heart Association Council on Nutrition, Physical Activity, and Metabolism: endorsed by the American College of Cardiology Foundation.” Circulation 2004; 110:2952.
-
Knoops K.T., de Groot L.C., Kromhout D., et al. “Mediterranean diet, lifestyle factors, and 10-year mortality in elderly European men and women: the HALE project.” JAMA 2004; 292:1433.
-
Kwaliteitsinstituut voor de gezondheidszorg CBO and Nederlandse Huisartsen Genootschap. (2006). Multidisciplinaire richtlijn cardiovasculair risicomanagement 2006. Alphen aan den Rijn: Van Zuiden Communications.
-
Lilly L.S. (ed.). (2007). Pathophysiology of Heart Disease. Baltimore: Lippincott Williams & Wilkins, p. 118–140.
-
Lilly L.S. (ed.). (2007). Pathophysiology of Heart Disease. Baltimore: Lippincott Williams & Wilkins, p. 118–140.
-
Nordmann A.J., Suter-Zimmermann K., Bucher H.C., et al. “Meta-analysis comparing Mediterranean to low-fat diets for modification of cardiovascular risk factors.” Am J Med 2011; 124:841.
-
Mancia G., Messerli F., Bakris G., et al. “Blood pressure control and improved cardiovascular outcomes in the International Verapamil SR-Trandolapril Study.” Hypertension 2007; 50:299.
-
McCully K.S. “Vascular pathology of homocysteinemia: implications for the pathogenesis of arteriosclerosis.” Am J Pathol 1969; 56:111.
-
Mendis S., Puska P., Norrving B. (eds.). (2011). Global Atlas on Cardiovascular Disease Prevention and Control. Geneva: World Health Organization.
-
Mukamal K.J., Chung H., Jenny N.S., et al. “Alcohol consumption and risk of coronary heart disease in older adults: the Cardiovascular Health Study.” J Am Geriatr Soc 2006; 54:30.
-
Newby D.E., Wright R.A., Labinjoh C., et al. “Endothelial dysfunction, impaired endogenous fibrinolysis, and cigarette smoking: a mechanism for arterial thrombosis and myocardial infarction.” Circulation 1999; 99:1411.
-
'Keefe O'Keefe J.H., Bybee K.A., Lavie C.J. “Alcohol and cardiovascular health: the razor-sharp double-edged sword.” J Am Coll Cardiol 2007; 50:1009.
-
Paffenbarger R.S. Jr, Hyde R.T., Wing A.L., et al. “The association of changes in physical-activity level and other lifestyle characteristics with mortality among men.” N Engl J Med 1993; 328:538.
-
Pasceri V., Cheng J.S., Willerson J.T., et al. “Modulation of C-reactive protein-mediated monocyte chemoattractant protein-1 induction in human endothelial cells by anti-atherosclerosis drugs.” Circulation 2001; 103:2531.
-
Pasceri V., Willerson J.T., Yeh E.T. “Direct proinflammatory effect of C-reactive protein on human endothelial cells.” Circulation 2000; 102:2165.
-
Pearson T.A., Mensah G.A., Alexander R.W., et al. “Markers of inflammation and cardiovascular disease: application to clinical and public health practice: A statement for healthcare professionals from the Centers for Disease Control and Prevention and the American Heart Association.” Circulation 2003; 107:499.
-
Poirier P., Giles T.D., Bray G.A., et al. “Obesity and cardiovascular disease: pathophysiology, evaluation, and effect of weight loss: an update of the 1997 American Heart Association Scientific Statement on Obesity and Heart Disease from the Obesity Committee of the Council on Nutrition, Physical Activity, and Metabolism.” Circulation 2006; 113:898.
-
Powell K.E., Thompson P.D., Caspersen C.J., Kendrick J.S. “Physical activity and the incidence of coronary heart disease.” Annu Rev Public Health 1987; 8:253.
-
Reaven G., Tsao P.S. “Insulin resistance and compensatory hyperinsulinemia: the key player between cigarette smoking and cardiovascular disease?” J Am Coll Cardiol 2003; 41:1044.
-
Ridker P.M., Cushman M., Stampfer M.J., et al. “Inflammation, aspirin, and the risk of cardiovascular disease in apparently healthy men.” N Engl J Med 1997; 336:973.
-
Ridker P.M., Glynn R.J., Hennekens C.H. “C-reactive protein adds to the predictive value of total and HDL cholesterol in determining risk of first myocardial infarction.” Circulation 1998; 97:2007.
-
Rimm E.B., Ascherio A., Giovannucci E., et al. “Vegetable, fruit, and cereal fiber intake and risk of coronary heart disease among men.” JAMA 1996; 275:447.
-
Rolland P.H., Friggi A., Barlatier A., et al. “Hyperhomocysteinemia-induced vascular damage in the minipig. Captopril-hydrochlorothiazide combination prevents elastic alterations.” Circulation 1995; 91:1161.
-
Ronksley P.E., Brien S.E., Turner B.J., et al. “Association of alcohol consumption with selected cardiovascular disease outcomes: a systematic review and meta-analysis.” BMJ 2011; 342:d671.
-
Rose G., Hamilton P.J., Colwell L., Shipley M.J. “A randomised controlled trial of anti-smoking advice: 10-year results.” J Epidemiol Community Health 1982; 36:102.
-
Rossouw J.E., Anderson G.L., Prentice R.L., et al. “Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results From the Women's Health Initiative randomized controlled trial.” JAMA 2002; 288:321.
-
Rozanski A., Blumenthal J.A., Kaplan J. “Impact of psychological factors on the pathogenesis of cardiovascular disease and implications for therapy.” Circulation 1999; 99:2192.
-
Rubin R., Strayer D.S., Rubin E. (ed.). (2008). Rubin's Pathology: Clinicopathologic Foundations of Medicine Fifth Edition. Baltimore: Lippincott Williams & Wilkins, p387-408
-
Sacks F.M., Pfeffer M.A., Moye L.A., Rouleau J.L., Rutherford J.D., Cole T.G., Brown L., Warnica J.W., Arnold J.M., Wun C.C., Davis B.R., Braunwald E. "The effect of pravastatin on coronary events after myocardial infarction in patients with average cholesterol levels. Cholesterol and Recurrent Events Trial investigators." N Engl J Med. 1996;335(14):1001.
-
Shepherd J., Cobbe S.M., Ford I., Isles C.G., Lorimer A.R., MacFarlane P.W., McKillop J.H., Packard C.J. "Prevention of coronary heart disease with pravastatin in men with hypercholesterolemia. West of Scotland Coronary Prevention Study Group." N Engl J Med. 1995;333(20):1301.
-
Stefanadis C., Tsiamis E., Vlachopoulos C., et al. “Unfavorable effect of smoking on the elastic properties of the human aorta.” Circulation 1997; 95:31.
-
Tall, A. R. "CETP inhibitors to increase HDL cholesterol levels." N.Engl.J.Med. 356.13 (2007): 1364-66.
-
The health benefits of smoking cessation. A report of the Surgeon General. DHHS Publication (CDC) 90-8416; U.S. Department of Health and Human Services, Washington, DC, 1990.
-
Theorell, T., Lind, E., Floderus, B. “The relationship of disturbing life-changes and emotions to the development of myocardial infarction and other serious diseases.” Int J Epidemiol 1975; 4:281.
-
Vita J.A., Keaney J.F. Jr. “Endothelial function: a barometer for cardiovascular risk? Circulation” 2002; 106:640.